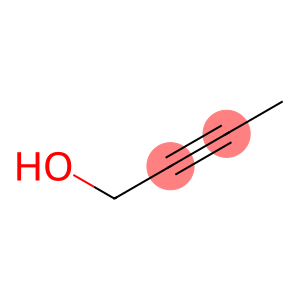
but-2-yn-1-ol
but-2-yn-1-ol
CAS: 764-01-2
Molecular Formula: C4H6O
but-2-yn-1-ol - Names and Identifiers
| Name | but-2-yn-1-ol |
| Synonyms | 2-Butynol 2-butyn-1-0l 2-Butyn-1-ol 2-BUTYNE-1-OL but-2-yn-1-ol 2-Butyne-1-ol BUT-2-IN-1-OL 2-Butynyl alcohol 1-hydroxy-2-butyne HYDROXYMETHYLMETHYLACETYLENE |
| CAS | 764-01-2 |
| EINECS | 212-113-4 |
| InChI | InChI=1/C4H6O/c1-2-3-4-5/h5H,4H2,1H3 |
but-2-yn-1-ol - Physico-chemical Properties
| Molecular Formula | C4H6O |
| Molar Mass | 70.09 |
| Density | 0.937 g/mL at 25 °C (lit.) |
| Melting Point | -2.2 °C (lit.) |
| Boling Point | 142-143 °C (lit.) |
| Flash Point | 125°F |
| Solubility | Miscible with chloroform and methanol. |
| Vapor Presure | 1.67mmHg at 25°C |
| Appearance | Liquid |
| Specific Gravity | 0.937 |
| Color | Clear colorless to light yellow |
| BRN | 1733676 |
| pKa | 13.14±0.10(Predicted) |
| Storage Condition | Refrigerator |
| Stability | Stable. Flammable. |
| Refractive Index | n20/D 1.453(lit.) |
but-2-yn-1-ol - Risk and Safety
| Risk Codes | R10 - Flammable R43 - May cause sensitization by skin contact R52/53 - Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S36/37 - Wear suitable protective clothing and gloves. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S16 - Keep away from sources of ignition. |
| UN IDs | UN 1987 3/PG 3 |
| WGK Germany | 3 |
| HS Code | 29052990 |
| Hazard Note | Irritant |
| Hazard Class | 3 |
| Packing Group | III |
but-2-yn-1-ol - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| Introduction | 2-butyne-1-ol is colorless or light yellow liquid at normal temperature and pressure, soluble in common organic solvents, the need for low temperature storage. 2-butyne-1-ol is a commonly used organic building block, which contains both unsaturated triple bond and alcohol hydroxyl group, and can realize the transformation of various functional groups. |
| uses | 2-butyne-1-ol can be used as a drug molecule and an intermediate in organic synthesis, in the conversion of organic synthesis, the hydroxyl group on the structure can be easily alkylated under alkaline conditions; The unsaturated triple bond can be carried out under the guidance of the hydroxyl group, halogen hydrogenation reaction and so on. |
| synthesis method | p-toluenesulfonic acid (3.30 g,17.4 mmol,0.1 eq) to a solution of THP-protected 2-butyne-1-ol (174 mmol) in methanol (solvent grade, 139 ml) was added and the reaction mixture was stirred overnight. Potassium carbonate (2.40g, 17.4 mmol, 0.1 EQ) was added to the reaction mixture and the resulting suspension was stirred for 30 min. The solid was removed by filtration and the solvent was evaporated under reduced pressure. The desired product was obtained by purification of the residue by distillation (76 mbar, oil bath temperature 120°C). |
Last Update:2024-04-09 20:49:11
Supplier List
Spot supply
Product Name: 2-Butyn-1-ol Visit Supplier Webpage Request for quotationCAS: 764-01-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 2-Butyn-1-ol Visit Supplier Webpage Request for quotationCAS: 764-01-2
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Multiple Specifications
Product Name: 2-Butyn-1-ol Request for quotationCAS: 764-01-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 2-Butyn-1-ol Visit Supplier Webpage Request for quotationCAS: 764-01-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 2-Butyn-1-ol Visit Supplier Webpage Request for quotationCAS: 764-01-2
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Multiple Specifications
Product Name: 2-Butyn-1-ol Request for quotationCAS: 764-01-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
View History


