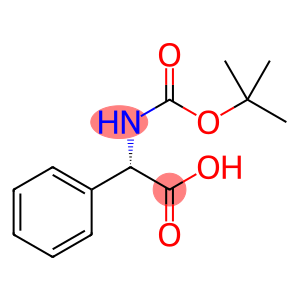
boc-L-alpha-phenylglycine
boc-L-alpha-phenylglycine
CAS: 2900-27-8
Molecular Formula: C13H17NO4
boc-L-alpha-phenylglycine - Names and Identifiers
| Name | boc-L-alpha-phenylglycine |
| Synonyms | Boc-Phg-OH Boc-L-Phg-OH Boc-L-phenylglycine N-Boc-L-phenylglycine (S)-Boc-L-Phenylglycine boc-L-alpha-phenylglycine (Tert-Butoxy)Carbonyl Phg-OH (S)-Boc-2-aminophenylacetic acid N-Boc-L-2-phenylglycineBoc-Phg-OH 2-(tert-butoxycarbonyl)-2-phenylacetic acid N-alpha-tert-Butoxycarbonyl-L-phenylglycine [(tert-butoxycarbonyl)amino](phenyl)acetic acid Boc-Phg-OH~N-(tert-Butoxycarbonyl)-L-phenylglycine (2S)-[(tert-butoxycarbonyl)aMino](phenyl)ethanoic acid (2S)-[(tert-butoxycarbonyl)amino](phenyl)ethanoic acid (2R)-[(tert-butoxycarbonyl)amino](phenyl)ethanoic acid (S)-2-((tert-Butoxycarbonyl)aMino)-2-phenylacetic acid |
| CAS | 2900-27-8 |
| EINECS | 000-000-0 |
| InChI | InChI=1/C13H17NO4/c1-13(2,3)18-12(17)14-10(11(15)16)9-7-5-4-6-8-9/h4-8,10H,1-3H3,(H,14,17)(H,15,16)/t10-/m0/s1 |
boc-L-alpha-phenylglycine - Physico-chemical Properties
| Molecular Formula | C13H17NO4 |
| Molar Mass | 251.28 |
| Density | 1.182±0.06 g/cm3(Predicted) |
| Melting Point | 88-91°C |
| Boling Point | 407.2±38.0 °C(Predicted) |
| Specific Rotation(α) | 142 ° (C=1, EtOH) |
| Flash Point | 200.1°C |
| Water Solubility | Insoluble in water |
| Vapor Presure | 2.32E-07mmHg at 25°C |
| Appearance | White crystal |
| Color | White to Almost white |
| BRN | 3592362 |
| pKa | 3.51±0.10(Predicted) |
| Storage Condition | Sealed in dry,2-8°C |
| Refractive Index | 142 ° (C=1, EtOH) |
| MDL | MFCD00065588 |
boc-L-alpha-phenylglycine - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| HS Code | 2924 29 70 |
boc-L-alpha-phenylglycine - Reference Information
| Application | N-(tert-butoxycarbonyl)-L-2-phenylglycine is an amino acid derivative, as an important raw material compound and drug intermediate, It can be used to synthesize β-lactam antibiotics such as penicillin, vilamycin, and prenamycin I , and can also be used for the synthesis of anti-tumor drug violet. N-(tert-butoxycarbonyl)-L-2-phenylglycine is its N-protected product and is widely used in pharmaceutical and chemical industries. |
| Synthesis method | Using L-2-phenylglycine as raw material, it reacts with di-tert-butyl dicarbonate under Et 3 N/MeOH conditions for Boc protection to prepare the target compound N-(tert-butoxycarbonyl)-L-2-phenylglycine. The preparation reaction formula of N-(tert-butoxycarbonyl)-L-2-phenylglycine is as follows: |
Last Update:2024-04-09 21:11:58
Supplier List
Spot supply
Product Name: Boc-Phg-OH Visit Supplier Webpage Request for quotationCAS: 2900-27-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: N-Boc-L-alpha-phenylglycine Visit Supplier Webpage Request for quotationCAS: 2900-27-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: BOC-D-PHG-OH Request for quotation
CAS: 2900-27-8
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2900-27-8
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Boc-L-α-phenylglycine Visit Supplier Webpage Request for quotationCAS: 2900-27-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Boc-Phg-OH Visit Supplier Webpage Request for quotationCAS: 2900-27-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: N-Boc-L-alpha-phenylglycine Visit Supplier Webpage Request for quotationCAS: 2900-27-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: BOC-D-PHG-OH Request for quotation
CAS: 2900-27-8
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 2900-27-8
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Spot supply
Product Name: Boc-L-α-phenylglycine Visit Supplier Webpage Request for quotationCAS: 2900-27-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



