benfluralin
benfluralin
CAS: 1861-40-1
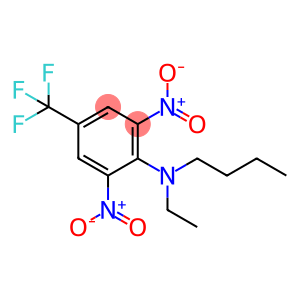
Molecular Formula: C13H16F3N3O4
benfluralin - Names and Identifiers
| Name | benfluralin |
| Synonyms | Binn EL-110 Benuralin benfluralin N-Butyl-2,6-dinitro-N-ethyl-4-trifluoromethylaniline N-butyl-N-ethyl-2,6-dinitro-4-(trifluoromethyl)aniline n-butyl-2,6-dinitro-n-ethyl-4-(trifluoromethyl)aniline N-Butyl-N-ethyl-2,6-dinitro-4-(trifluoromethyl)benzenamine n-butyln-ethyl-2,6-dinitro-4-(trifluoromethyl)benzeneamine n-butyl-n-ethyl-2,6-dinitro-4-(trifluoromethyl)-benzenamin N-butyl-N-ethyl-alpha,alpha,alpha-trifluoro-2,6-dinitro-p-toluidine |
| CAS | 1861-40-1 |
| EINECS | 217-465-2 |
| InChI | InChI=1/C13H16F3N3O4/c1-3-5-6-17(4-2)12-10(18(20)21)7-9(13(14,15)16)8-11(12)19(22)23/h7-8H,3-6H2,1-2H3 |
benfluralin - Physico-chemical Properties
| Molecular Formula | C13H16F3N3O4 |
| Molar Mass | 335.28 |
| Density | 1.3435 (estimate) |
| Melting Point | 65-66.5° |
| Boling Point | 121~122℃ (0.5mmHg) |
| Flash Point | 177°C |
| Water Solubility | <1mg/L(25 ºC) |
| Vapor Presure | 1.21E-05mmHg at 25°C |
| Appearance | Crystalline Solid |
| Color | Yellow-orange |
| Merck | 13,1037 |
| pKa | -0.59±0.50(Predicted) |
| Storage Condition | 0-6°C |
| Refractive Index | 1.527 |
benfluralin - Risk and Safety
| Hazard Symbols | N - Dangerous for the environment |
| Risk Codes | 50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 3077 |
| RTECS | XU4550000 |
| HS Code | 29214300 |
| Toxicity | LD50 orally in female rats: >10,000 mg/kg (Goldenthal) |
benfluralin - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| properties | fluoxalamine has a melting point of 65~66.5 ℃, a vapor pressure of 0.533 × 10-4Pa(25 ℃), slightly soluble in water (70mg/l at 25 ℃), and easily soluble in organic solvents. |
| Synthesis method | Using p-chlorotoluene as raw material, it reacts with chlorine under light to generate p-chloro-trichloromethyl benzene, reacts with hydrogen fluoride to generate p-chloro-trifluoromethyl benzene, nitrification in sulfuric acid solution to generate 2, 6-dinitro-4-trifluoromethyl chlorobenzene, in sodium hydroxide solution, it reacts with N-ethyl-N-butyl amine to produce fluorosamine [1]. |
| use | phenfluralin is a pre-bud herbicide used to control grass and other weeds in a range of edible and non-edible crops. It has low water solubility and is very volatile. It is not expected to seep into groundwater. It is moderately persistent in soil systems and usually not persistent in water. Its toxicity to mammals is low, but there are concerns about its bioaccumulation potential. It is moderately toxic to birds, bees, earthworms and most aquatic organisms. Fluoxamine can be used as a herbicide, weeding before budding, and can control annual narrow leaves and broad-leaved weeds. Fluoxalamine is a dinitroaniline herbicide used to control the growth of weeds. In 2004, the use in the United States was about 700,000 pounds. |
| Suitable for crops | Stellaria; Sheep shed; Portulaca oleracea; Polygonum cuspidatum; Clover; Chenopodium; Plagella; Turf; Lettuce; Some specialty crops, including chicory; Ornaments, including Christmas trees; Fruitless fruit trees and nut trees. |
| biological activity | Benfluralin is a herbicide and chemical pesticide, which can be used as an early-maturing herbicide to control weeds in a series of food and non-food crops. |
| Production method | Produced by the reaction of 4-chloro-3, 5-dinitrotrifluorotoluene and ethylbutylamine. |
| category | pesticide |
| toxicity classification | poisoning |
| acute toxicity | oral-rat LD50: 10000 mg/kg; Oral-mouse LD50: 5000 mg/kg |
| flammability hazard characteristics | Combustion produces toxic nitrogen oxides and fluoride gases |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; separate from food raw materials storage and transportation |
| fire extinguishing agent | dry powder, foam, sand |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 21:21:28
Supplier List
Spot supply
Product Name: Benfluralin Visit Supplier Webpage Request for quotationCAS: 1861-40-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Benfluralin Request for quotation
CAS: 1861-40-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1861-40-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Multiple Specifications
Product Name: Benfluralin Visit Supplier Webpage Request for quotationCAS: 1861-40-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Benfluralin Visit Supplier Webpage Request for quotationCAS: 1861-40-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Benfluralin Request for quotation
CAS: 1861-40-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1861-40-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Multiple Specifications
Product Name: Benfluralin Visit Supplier Webpage Request for quotationCAS: 1861-40-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


