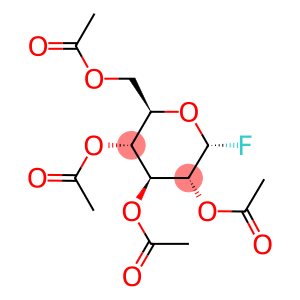
acetofluoro-A-D-glucose
acetofluoro-A-D-glucose
CAS: 3934-29-0
Molecular Formula: C14H19FO9
acetofluoro-A-D-glucose - Names and Identifiers
| Name | acetofluoro-A-D-glucose |
| Synonyms | acetofluoro-A-D-glucose Tetraacetyl-alpha-D-glucose fluoride a-D-Glucopyranosyl fluoride tetraacetate alpha-D-Glucopyranosyl fluoride tetraacetate Tetra-O-acetyl-alpha-D-glucopyranosyl fluoride a-D-Glucopyranosyl fluoride,2,3,4,6-tetraacetate 2,3,4,6-Tetra-O-Acetyl-Alpha-D-Glucopyranosyl Fluoride 2,3,4,6-TETRA-O-ACETYL-ALPHA-D-GLUCOPYRANOSYL FLUORIDE (EP) |
| CAS | 3934-29-0 |
| InChI | InChI=1/C14H19FO9/c1-6(16)20-5-10-11(21-7(2)17)12(22-8(3)18)13(14(15)24-10)23-9(4)19/h10-14H,5H2,1-4H3/t10-,11-,12+,13-,14+/m1/s1 |
acetofluoro-A-D-glucose - Physico-chemical Properties
| Molecular Formula | C14H19FO9 |
| Molar Mass | 350.29 |
| Density | 1.30 |
| Melting Point | 105.0 to 109.0 °C |
| Boling Point | 374.8±42.0 °C(Predicted) |
| Flash Point | 174.346°C |
| Solubility | soluble in Chloroform |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Almost white |
| Storage Condition | Sealed in dry,2-8°C |
| Refractive Index | 1.465 |
| MDL | MFCD00792705 |
acetofluoro-A-D-glucose - Risk and Safety
| WGK Germany | 3 |
| HS Code | 29329990 |
acetofluoro-A-D-glucose - Introduction
acetofluoro-A-D-glucose (acetofluoro-α-D-glucose) is an organic compound whose chemical formula is C10H14O6F.
Nature:
-Appearance: Colorless solid
-Solubility: Soluble in organic solvents, such as methanol, dimethyl sulfoxide, slightly soluble in water
-melting point: about 160-165 ℃
-relative molecular mass: 270.22g/mol
Use:
-Chemical research: It can be used as an intermediate in organic synthesis and used in the synthesis of organic compounds, such as drugs, pesticides, dyes, etc.
-Medical field: It may have anti-diabetic, anti-viral and anti-tumor activities, so it also has potential application value in medical research.
Preparation Method:
There are many ways to prepare this compound. Common methods include:
1. The glucopyranose is reacted with an acetylating agent (such as acetic anhydride) under acidic conditions to generate 2,3,4,6-tetra-O-acetyl-α-D-glucopyranose.
2. Next, the obtained acetylated product is reacted with a fluorinating agent such as stannous fluoride to replace the oxygen atom (O) at the position of the hydroxyl group (OH) therein with a fluorine atom (F), thereby obtaining a acetofluoro-A-D-glucose.
Safety Information:
-The toxicity and irritation data of this compound are limited, so it is necessary to follow general laboratory safety practices.
-If the compound needs to be added to a product or drug, corresponding safety evaluation and testing are required to ensure the safety of use.
Please note that this compound is only used for research and experimental purposes and should be used in accordance with relevant laws and regulations and in a safe environment.
Nature:
-Appearance: Colorless solid
-Solubility: Soluble in organic solvents, such as methanol, dimethyl sulfoxide, slightly soluble in water
-melting point: about 160-165 ℃
-relative molecular mass: 270.22g/mol
Use:
-Chemical research: It can be used as an intermediate in organic synthesis and used in the synthesis of organic compounds, such as drugs, pesticides, dyes, etc.
-Medical field: It may have anti-diabetic, anti-viral and anti-tumor activities, so it also has potential application value in medical research.
Preparation Method:
There are many ways to prepare this compound. Common methods include:
1. The glucopyranose is reacted with an acetylating agent (such as acetic anhydride) under acidic conditions to generate 2,3,4,6-tetra-O-acetyl-α-D-glucopyranose.
2. Next, the obtained acetylated product is reacted with a fluorinating agent such as stannous fluoride to replace the oxygen atom (O) at the position of the hydroxyl group (OH) therein with a fluorine atom (F), thereby obtaining a acetofluoro-A-D-glucose.
Safety Information:
-The toxicity and irritation data of this compound are limited, so it is necessary to follow general laboratory safety practices.
-If the compound needs to be added to a product or drug, corresponding safety evaluation and testing are required to ensure the safety of use.
Please note that this compound is only used for research and experimental purposes and should be used in accordance with relevant laws and regulations and in a safe environment.
Last Update:2024-04-09 20:49:11
Supplier List
Product Name: 2,3,4,6-TETRA-O-ACETYL-ALPHA-D-GLUCOPYRANOSYL FLUORIDE Request for quotation
CAS: 3934-29-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3934-29-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2,3,4,6-Tetra-O-acetyl-α-D-glucopyranosyl fluoride Visit Supplier Webpage Request for quotationCAS: 3934-29-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3,4,6-Tetra-o-Acetyl-Alpha-D-Glucopyranosyl Fluoride Visit Supplier Webpage Request for quotation
CAS: 3934-29-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3934-29-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2,3,4,6-TETRA-O-ACETYL-ALPHA-D-GLUCOPYRANOSYL FLUORIDE Request for quotation
CAS: 3934-29-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3934-29-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 2,3,4,6-Tetra-O-acetyl-α-D-glucopyranosyl fluoride Visit Supplier Webpage Request for quotationCAS: 3934-29-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3,4,6-Tetra-o-Acetyl-Alpha-D-Glucopyranosyl Fluoride Visit Supplier Webpage Request for quotation
CAS: 3934-29-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3934-29-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


