Triforine
Triforine
CAS: 26644-46-2
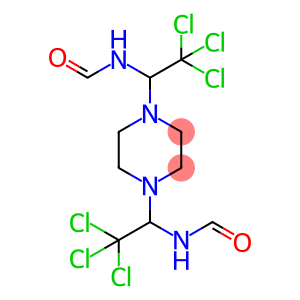
Molecular Formula: C10H14Cl6N4O2
Triforine - Names and Identifiers
| Name | Triforine |
| Synonyms | SAPROL TRIFORIN Triforine TRIFORINE SAPROL(R) 1,4-bis(1-formamido-2,2,2-trichloroethyl)piperazine 1,4-bis(1-formamido-2,2,2-trichloroethyl)-piperazin PIPERAZIN-1,4-DIYL-BIS-[1-(2,2,2-TRICHLOROETHYL)FORMAMIDE] N,N'-[PIPERAZINE-1,4-DIYLBIS(TRICHLOROMETHYL)METHYLENE]-DIFORMAMIDE |
| CAS | 26644-46-2 |
| EINECS | 247-872-0 |
| InChI | InChI=1/C10H14Cl6N4O2/c11-9(12,13)7(17-5-21)19-1-2-20(4-3-19)8(18-6-22)10(14,15)16/h5-8H,1-4H2,(H,17,21)(H,18,22) |
Triforine - Physico-chemical Properties
| Molecular Formula | C10H14Cl6N4O2 |
| Molar Mass | 434.96 |
| Density | 1.5269 (rough estimate) |
| Melting Point | 155℃ (decomposition) |
| Boling Point | 561.5±50.0 °C(Predicted) |
| Flash Point | 293.4°C |
| Water Solubility | 9 mg l-1 (20 °C) |
| Solubility | DMSO (Slightly) |
| Vapor Presure | 8 x 10-2 Pa (25 °C) |
| Appearance | neat |
| Color | White to Off-White |
| Merck | 13,9762 |
| BRN | 626358 |
| pKa | 10.6 (base) |
| Storage Condition | 0-6°C |
| Refractive Index | 1.6000 (estimate) |
| Physical and Chemical Properties | Pure white crystals, m. P. 155 °c, vapor pressure 2.67 x 10-8Pa (25 °c). Room temperature solubility: dimethylformamide 28.3g/L, methanol 1.13g/L, dioxane 1.66g/L, toluene 0.88g/L, slightly soluble in acetone, benzene, carbon tetrachloride, chloroform, dichloromethane, difficult to dissolve in dimethyl sulfoxide, water solubility of 27~29mg/L. |
Triforine - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R52/53 - Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment. R20 - Harmful by inhalation |
| Safety Description | 61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN3077 (solid); UN3082 (liquid) |
| WGK Germany | 2 |
| RTECS | TK9200000 |
| Toxicity | LD50 oral in rat: 6gm/kg |
Triforine - Reference Information
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| Properties | Zimalin, also known as Zimalin, is a heterocyclic fungicide. The original drug is white and tasteless, almost insoluble in water, alkali and acid. Zimalin has little solubility in chloroform and aromatic hydrocarbons, and is slightly soluble in methanol and ketones. The solubility of zimalin in dimethylformamide and N-methylpyrrolidone is 30% and 40%. |
| application | zimalin is a systemic fungicide, which is used to control cereal rust caused by powdery mildew, rust fungus, black star fungus, etc. and apple black star disease, cereals, apples, cucurbitaceae powdery mildew and other diseases. The effect of watering the roots is better than spraying on the leaves, and it is also used for seed dressing of cereal crops. Its effect on bacteria is to inhibit the biosynthesis of sterols and interfere with the formation of membranes. It damages the powdery mildew sucker in the plant body without affecting the germination of the spores. |
| toxicity | rats and mice acute oral LD50>6000mg/kg, rats and mice acute percutaneous> 5800mg/kg. Slight irritation to skin and eyes. Carp LC50>40mg/L (48h), Daphnia LC5040mg/L (48h), quail acute oral LD50>6000mg/kg, safe for bees. |
| use | piperazine systemic fungicides. It is mainly used to prevent and control powdery mildew and rust of vegetables, fruit trees and grains. When the content is 0.02% ~ 0.025%, it can effectively prevent and control powdery mildew, scab and other diseases of fruits and berries. When the content is 0. When 015%, it can prevent ornamental plants powdery mildew, rust and black spot; when the use content is 0.025%, it can prevent vegetable powdery mildew and other diseases. Grain powdery mildew can be controlled at a dose of 2~2.5g/100m2, while grain rust and fruit storage diseases can be controlled at a dose of 3g/100m2. |
| production method | trichloroacetaldehyde and formamide are mixed, heated for 1.5~2h under stirring, cooled, and chloral formamide crystals are obtained. Then add thionyl chloride, reflux for 30min, remove excess thionyl chloride after the reaction, the residue is dissolved in ether, the ether solution is washed with cold water, anhydrous magnesium sulfate is dried, and N-(1,2,2, 2-tetrachloroethyl) formamide is obtained after concentration. Dissolve the intermediate product in acetone, add the acetone solution of piperazine and triethylamine dropwise, stir for 2 hours at room temperature, inject the product into cold water, filter, recrystallize dioxane, deazinamine, m.p.175 ℃. mix trichloraldehyde with formaldehyde amine, heat for 1.5-2 hours under stirring, cool to room temperature, and all crystallize into chloral formamide (melting point 117-118 ℃). Add it to thionyl chloride, heat and reflux for half an hour, remove excess thionyl chloride after the reaction, dissolve the residue in ether, wash the ether liquid with cold water, dry with anhydrous magnesium sulfate, and concentrate to obtain N-(1,2,2 ',2 ''-tetrachloroethyl) formamide (melting point 94-95 ℃). Then dissolve in acetone, drop the acetone solution of piperazine and triethylamine, stir for 2 hours at room temperature, inject the product into cold water, filter, and crystallize with dioxane to obtain ammonia. Raw material consumption quota: chloroacetaldehyde 880kg/t, formamide 270kg/t, thionyl chloride 1060kg/t, N-(1,2,2,2-tetrachloroethyl) formamide 1130kg/t, piperazine 230kg/t, triethylamine 560kg/t. |
| category | pesticide |
| toxicity classification | low toxicity |
| acute toxicity | oral-rat LD50: 6000 mg/kg; Oral-mouse LD50: 6000 mg/kg |
| flammability hazard characteristics | combustion produces toxic chloride and nitrogen oxide gas |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; separate from food raw materials storage and transportation |
| fire extinguishing agent | dry powder, foam, sand |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Triforin Visit Supplier Webpage Request for quotationCAS: 26644-46-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TRIFORINE Request for quotation
CAS: 26644-46-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 26644-46-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Multiple Specifications
Product Name: Triforine (mixture of isomers) Visit Supplier Webpage Request for quotationCAS: 26644-46-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Triforin Visit Supplier Webpage Request for quotationCAS: 26644-46-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TRIFORINE Request for quotation
CAS: 26644-46-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 26644-46-2
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
Multiple Specifications
Product Name: Triforine (mixture of isomers) Visit Supplier Webpage Request for quotationCAS: 26644-46-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


