Ranitidinebismuthcitrate
Ranitidinebismuthcitrate
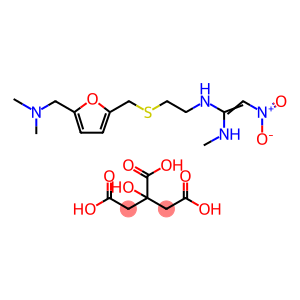
CAS: 128345-62-0
Molecular Formula: C19H30N4O10S
Ranitidinebismuthcitrate - Names and Identifiers
| Name | Ranitidinebismuthcitrate |
| Synonyms | Pylorid Gastrimuto Ranitidine bismutrex RANTIDINEBISMUTHCITRATE Ranitidinebismuthcitrate Ranitidine Bismuth Citrate TIANFU CHEM-- Ranitidinebismuthcitrate 2-Hydroxy-1,2,3-propanetricarboxylic acid bismuth(3+) salt compd with N-[2-[[[5-[(dimethylamino)methyl]-2-furanyl]methyl]thio]ethyl]-N'-methyl-2-nitro-1,1-ethenediamine |
| CAS | 128345-62-0 |
| EINECS | 1308068-626-2 |
| InChI | InChI=1/C13H22N4O3S.C6H8O7.Bi/c1-14-13(9-17(18)19)15-6-7-21-10-12-5-4-11(20-12)8-16(2)3;7-3(8)1-6(13,5(11)12)2-4(9)10;/h4-5,9,14-15H,6-8,10H2,1-3H3;13H,1-2H2,(H,7,8)(H,9,10)(H,11,12);/q;;+3/p-3/b13-9-; |
Ranitidinebismuthcitrate - Physico-chemical Properties
| Molecular Formula | C19H30N4O10S |
| Molar Mass | 506.53 |
| Boling Point | 437.1°C at 760 mmHg |
| Flash Point | 218.2°C |
| Vapor Presure | 7.66E-08mmHg at 25°C |
Ranitidinebismuthcitrate - Standard
Authoritative Data Verified Data
This product is ranitidine and bismuth citrate to form a complex of variable composition. According to the dry product, ranitidine and bismuth citrate content of 1:1, containing ranitidine (C13H22N403S) should be 42.5% ~ 45.5%; Ranitidine and bismuth citrate content of 1:1.1, containing ranitidine (C13H22N403S) should be 39.5% to 42.5%; Bismuth citrate containing bismuth (Bi) calculated should be 27.5% to 30.5%.
Ranitidinebismuthcitrate - Trait
Authoritative Data Verified Data
- This product is white to light yellow-brown powder, or crystalline or granular powder; Deliquescence, color darkening after moisture absorption.
- This product is very soluble in water, and almost insoluble in ethanol, ether, acetone or chloroform.
Ranitidinebismuthcitrate - Differential diagnosis
Authoritative Data Verified Data
- take about 0.5g of this product, put it in a test tube, and slowly heat it with small fire. The gas generated can make the wet lead acetate test paper black.
- in the chromatogram recorded under the item of ranitidine content determination, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the control solution.
- take this product and make a solution containing about 25ug per 1 ml with water. According to ultraviolet-visible spectrophotometry (General 0401), there is maximum absorption at wavelengths of 228nm and 314nm.
- the aqueous solution of this product shows the identification reaction of bismuth salt (2) and citrate (General rule 0301).
Ranitidinebismuthcitrate - Exam
Authoritative Data Verified Data
acidity
take this product L. 0g, Add 10ml of water to dissolve, according to the law to determine (General 0631) ,pH value should be 4.5~6.5.
clarity of the solution
take l.Og of this product, Add 10ml of water to dissolve, the solution should be clear; If it is turbid, it should not be more concentrated compared with the No. 2 Turbidity standard solution (General rule 0902 first method).
Related substances
take an appropriate amount of this product (about equivalent to ranitidine lOOmg), put it in a 100ml measuring flask, add water to dissolve and dilute to the scale, shake well, and use it as a test solution, set in a 100ml measuring flask, dilute to the scale with water, and shake to serve as a control solution. According to the chromatographic conditions of ranitidine under the item of content determination, 10 u1 of control solution and 10 u1 of test solution are accurately measured, and human liquid chromatograph is injected respectively to record the chromatogram. If there are impurity peaks in the chromatogram of the test solution, the chromatographic peaks before the relative retention time of 0.15 shall be subtracted, and the area of a single impurity peak shall not be greater than the area of the main peak of the control solution (1.0% ) , the sum of each impurity peak area shall not be greater than 2 times (2.0%) of the main peak area of the control solution. The chromatogram of the test solution is 0.05 times smaller than the main peak area of the control solution.
sulfate
take 1.0g of this product, add 20ml of water to dissolve, add 4ml of dilute hydrochloric acid, shake, filter, filtrate is divided into 2 equal parts; Add 5ml of 25% barium chloride solution in 1 part, place for 10 minutes, filter repeatedly until the filtrate is clear, dilute to 40ml with water, add 2.0ml of standard potassium sulfate solution, add appropriate amount of water to make it 50ml, shake well, and place for 10 minutes, as a control solution; after the other portion was diluted to 40ml with water, 5ml of 25% barium chloride solution was added, and then an appropriate amount of water was added to make it 50ml. The mixture was shaken well and left for 10 minutes. Compared with the control solution, it should not be more concentrated (0.04%).
nitrate
take 0.50g of this product, put it in a test tube, add 5ml of water to dissolve, add 5ml of sulfuric acid, mix well, cool, slowly add 5ml of ferrous sulfate test solution along the tube wall to make two layers, contact surfaces should not be immediately Brown.
loss on drying
taking this product, using phosphorus pentoxide as desiccant, drying under reduced pressure at 60°C for 4 hours, the weight loss shall not exceed 6.0% (General rule 0831).
lead salt
take 1.0g of this product, burn at 600°C to make it ash completely. After cooling, add 0.5~1 ml of nitric acid Dropwise to dissolve, add potassium hydroxide solution (1-6) about 5ml, so that the pH value reaches 10 or more, boil for 2 minutes, let cool, filter, wash the residue with a small amount, wash and filtrate, adjust the pH value to 7 with acetic acid, dilute to 25ml with water, add 2ml of acetate buffer (pH 3.5) and 2ml of thioacetamide solution, shake well, and place for 2 minutes for color development, it should not be deeper (0.002%) compared with ml of standard lead solution, which was prepared by the same method.
Ranitidinebismuthcitrate - Content determination
Authoritative Data Verified Data
bismuth
take about 0.6g of this product, weigh accurately, add 50ml of water, shake to dissolve, add 3ml of nitric acid solution (l-3) and 2 drops of Xylenol Orange indicator solution, titrate with ethylene diamine tetraacetic acid disodium titration solution (0.05mol/L) until the solution yellow. Each 1 ml of ethylene diamine tetraacetic acid disodium titration solution (0.05mol/L) is equivalent to 10.45mg of Bi.
ranitidine
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
silica gel bonded with octa-alkyl silane as A filler (krolmasil C18,4.6mm X 150mm, 5um or equivalent column); Mobile phase A was phosphate buffer (6.8 of phosphoric acid, add 50% of 8.6 sodium hydroxide solution to 1900ml of water, add water to 50%, adjust pH to 7.1±0.05 with phosphoric acid or sodium hydroxide solution-acetonitrile (98:2), the mobile phase B was phosphate buffer-acetonitrile (78:22); The gradient elution was carried out as follows; The detection wavelength was 230nm. The flow rate was 1.5 per minute; The column temperature was 35°C. Take about 0.lg ranitidine hydrochloride, put it in a 100ml measuring flask, add 1ml of 50% sodium hydroxide solution, add about 60ml of water, shake to dissolve, dilute to the scale with water, shake well, and place it for 1 hour, 10UL was injected into the liquid chromatograph and the chromatogram was recorded. Adjust the flow rate or mobile phase ratio, so that the retention time of the main component chromatographic peak is about 12 minutes, the retention time of the ranitidine hydrochloride impurity I peak relative to the ranitidine peak is about 0.85, the resolution of ranitidine peak and ranitidine hydrochloride impurity I peak should be greater than 4.0.
assay
take an appropriate amount of this product (about 20mg equivalent to ranitidine), accurately weigh it, put it in a 200ml measuring flask, add water to dissolve and dilute to the scale, shake well, and use it as a test solution, A 10ul injection liquid chromatograph was used for precise measurement, and the chromatogram was recorded. Another 22mg of ranitidine hydrochloride reference substance was used for precise weighing, and the same method was used for determination. According to the external standard method to calculate the peak area, and the result is multiplied by 0.8961, that is.
Ranitidinebismuthcitrate - Category
Authoritative Data Verified Data
H2 receptor antagonist.
Ranitidinebismuthcitrate - Storage
Authoritative Data Verified Data
It was shielded from light, sealed, and stored in a cool-dark dry place.
Ranitidinebismuthcitrate - Ranitidine Bismuth Citrate Tablets
Authoritative Data Verified Data
This product is a complex of bismuth citrate and ranitidine. The amount of ranitidine and bismuth citrate is 1:1.1, and each tablet contains bismuth citrate calculated as bismuth (Bi), which should be 52.0 ~ 64.omg; Ranitidine containing (C13H22N403S) should be 72.0 ~ 88.omg.
trait
This product is a film-coated tablet, white to yellowish after removing the coating.
identification
- take an appropriate amount of fine powder of this product (about 0.5g equivalent to ranitidine bismuth citrate), place it in a test tube, and slowly heat it with small fire. The generated gas can make the wet lead acetate test paper black.
- in the chromatogram recorded under the content determination of ranitidine, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the control solution.
- take an appropriate amount of fine powder of this product, add water, shake, filter, the filtrate shows the identification reaction of bismuth salt (2) and citrate salt (2) (General rule 0301).
examination
- Related substances take an appropriate amount of fine powder of this product (about equivalent to ranitidine lOOmg ) , put it in a 100ml measuring flask, add an appropriate amount of water, fully shake to dissolve ranitidine, and dilute it with water to the scale, shake well, filter, and take the continued filtrate as a test solution; Take 1ml with precision, put it in a 100ml measuring flask, dilute it with water to the scale, shake well, and use it as a control solution. Determination of related substances of ranitidine Bismuth Citrate by this method. If there are impurity peaks in the chromatogram of the test solution, the chromatographic peaks before the relative retention time of 0.15 shall be subtracted, and the area of a single impurity peak shall not be greater than the area of the main peak of the control solution (1.0% ) , the sum of each impurity peak area shall not be greater than 2 times (2.0%) of the main peak area of the control solution. The chromatogram of the test solution is 0.05 times smaller than the main peak area of the control solution.
- the dissolution of this product, according to the dissolution and release determination method (General rule 0931 first method), with water as the dissolution medium, the speed is 100 rpm, according to the law, after 30 minutes, take the appropriate amount of the solution, filter it, take 5ml of the filtrate accurately, put it in a 50ml measuring flask, dilute it with water to the scale, shake it well, the absorbance was measured at a wavelength of 0401 NM according to ultraviolet-visible spectrophotometry (General rule 495), and the elution amount of each tablet was calculated as the absorption coefficient of C13H22N403S was. The limit is 80% of the labeled amount of ranitidine and should be met.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
- Take 20 tablets of bismuth, accurately weigh, grind, accurately weigh an appropriate amount (about 0.6g equivalent to ranitidine bismuth citrate), add 50ml of water, and fully shake to dissolve ranitidine bismuth citrate, add 3ml of nitric acid solution (1-3) and 2 drops of Xylenol Orange indicator solution, and titrate with ethylenediamine tetraacetic acid disodium titration solution (0.05mol/L) until the solution is yellow. Each 1 ml of ethylene diamine tetraacetic acid disodium titration solution (0.05mol/L) is equivalent to 10.45mg of Bi.
- ranitidine was measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica gel as filler (KR masil C18,4.6mm X 150mm, 5um or equivalent chromatographic column mobile phase A is phosphate buffer (take phosphoric acid 6.8, add 50% ml of 8.6 sodium hydroxide solution to 1900ml of water, add water to 50% ml, adjust pH to 7.1±0.05 with phosphoric acid or sodium hydroxide solution-acetonitrile (98:2 ), mobile Phase B was phosphate buffer-acetonitrile (78:22 ) ; Gradient elution was carried out as follows; Detection wavelength was 230um; Flow rate was 1.5ml per minute; Column temperature was 35°C. Take about 0.lg ranitidine hydrochloride, put it in a 100ml measuring flask, add 1ml of 50% sodium hydroxide solution, add about 60ml of water, shake to dissolve, dilute to the scale with water, shake well, and place it for 1 hour, 10u1 was injected into human liquid chromatograph, and the chromatogram was recorded. Adjust the flow rate or mobile phase ratio, so that the retention time of the main component chromatographic peak is about 12 minutes, the retention time of the ranitidine hydrochloride impurity I peak relative to the ranitidine peak is about 0.85, the resolution of ranitidine peak and ranitidine hydrochloride impurity I peak should be greater than 4.0.
- determination method an appropriate amount of fine powder (about equivalent to 20mg of ranitidine) under the item of bismuth content determination shall be accurately weighed and placed in a 200ml measuring flask, and an appropriate amount of water shall be added, fully shake to dissolve ranitidine and dilute to the scale with water, shake, filter, take the filtrate as the test solution, and inject 10u1 into the liquid chromatograph to record the chromatogram; another 22mg of ranitidine hydrochloride reference, precision weighing, dissolved in water and diluted to make each lml containing 0.llmg control solution, the same method. According to the external standard method to calculate the peak area, and the result is multiplied by 0.8961, that is.
category
Same as ranitidine bismuth citrate.
specification
0.2g (ranitidine and bismuth citrate 1:1.1)
storage
light-shielded, sealed, and stored in a dry place.
Ranitidinebismuthcitrate - Ranitidine bismuth citrate capsules
Authoritative Data Verified Data
This product contains ranitidine bismuth citrate complex. If the amount of ranitidine and bismuth citrate is 1:1.1, the content of ranitidine (C13H22N403S) per capsule should be 72.0 ~ mg, and the content of bismuth (Bi) is calculated as bismuth (Bi), should be 52.0 ~ 64.omg; Ranitidine and bismuth citrate amount of 1:1, each containing ranitidine (C13H22N403S) should be 139~169mg, bismuth citrate containing bismuth (Bi) calculation, should be 91 ~ lllmg.
trait
The contents of this product are yellowish to yellowish brown granules or powder.
identification
- take an appropriate amount of the content of this product (about 0.5g equivalent to ranitidine bismuth citrate), put it in a test tube, and slowly heat it with a small fire. The gas generated can make the wet lead acetate test paper black.
- in the chromatogram recorded under the item of ranitidine content determination, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the control solution.
- take an appropriate amount of the contents of this product, add water to shake, filter, and the filtrate shows the identification reaction of bismuth salt (2) and citrate salt (2) (General rule 0301).
examination
- Related substances take an appropriate amount of the contents of this product (about equivalent to ranitidine lOOmg), put it in a 100ml measuring flask, add water to dissolve ranitidine and dilute it to the scale, shake it well and filter it, the filtrate was taken as a test solution; 1 ml was accurately measured, placed in a 100ml measuring flask, diluted with water to scale, and shaken to serve as a control solution. According to the chromatographic conditions of ranitidine under the content determination item, l0ul of the control solution and the test solution are accurately measured, and the human liquid chromatograph is injected respectively, and the chromatogram is recorded. If there are impurity peaks in the chromatogram of the test solution, the chromatographic peaks before the relative retention time of 0.15 shall be subtracted, and the area of a single impurity peak shall not be greater than the area of the main peak of the control solution (1.0%), the sum of each impurity peak area shall not be greater than 2 times (2.0%) of the main peak area of the control solution. The chromatogram of the test solution is smaller than the pair, and the chromatographic peak with the main peak area of 0.05 times of the solution is ignored.
- others should comply with the relevant provisions under the capsule (General 0103).
Content determination
- the contents under the content difference item of bismuth loading were mixed evenly, and an appropriate amount (equivalent to ranitidine bismuth citrate 0.6g) was accurately weighed, 50ml of water was added, and the bismuth citrate was dissolved by fully shaking, 3ml of nitric acid solution (l-3) and 2 drops of Xylenol Orange indicator solution were added, and the solution was titrated with ethylenediamine tetraacetic acid disodium titration solution (0.05mol/L) to show yellow color. Each 1 ml of ethylene diamine tetraacetic acid disodium titration solution (0.05mol/L) is equivalent to 10.45mg of Bi.
- ranitidine was measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica gel as the filler (krolmasil C18,4.6mm X 150mm, 5um or performance equivalent column); the mobile phase A was phosphate buffer (6.8 of phosphoric acid was taken, 1900ml of water was placed, 50% of 8.6 sodium hydroxide solution was added, and water was added to 50%, and the pH value was adjusted to 7.1±0.05 with phosphoric acid or sodium hydroxide solution).-Acetonitrile (98:2 ) , mobile phase B was phosphate buffer-acetonitrile (78:22); Gradient elution was carried out according to the following table; Detection wavelength was 230nm. The flow rate was 1.5 per minute; The column temperature was 35°C;. Take ranitidine hydrochloride about O.lg, put in a 100ml measuring flask, add 1 ml of 50% sodium hydroxide solution, add about 60ml of water, shake to dissolve, dilute with water to the scale, shake well, and place for 1 hour, 10UL was injected into the liquid chromatograph and the chromatogram was recorded. Adjust the flow rate or mobile phase ratio, so that the retention time of the main component chromatographic peak is about 12 minutes, the retention time of the ranitidine hydrochloride impurity I peak relative to the ranitidine peak is about 0.85, the resolution of ranitidine peak and ranitidine hydrochloride impurity I peak should be greater than 4.0.
- determine the contents under the item of loading difference, mix evenly, accurately weigh an appropriate amount (about 20mg equivalent to ranitidine), put it in a 200ml measuring flask, and add an appropriate amount of water, fully shake to dissolve ranitidine and dilute to the scale with water, shake well, filter, take the filtrate as the test solution, inject 10ul into the liquid chromatograph with precision, record the chromatogram; another 22mg of ranitidine hydrochloride reference, precision weighing, dissolved in water and diluted to make each lml containing 0.llmg control solution, the same method. According to the external standard method to calculate the peak area, and the result is multiplied by 0.8961, that is.
category
Same as ranitidine bismuth citrate.
specification
- 0.2g (ranitidine and bismuth citrate 1:1.1)
- 0.35g (ranitidine and bismuth citrate 1:1)
storage
sealed and stored in a dry place.
Ranitidinebismuthcitrate - Reference Information
| overview | bismuth citrate ranitidine (RBC) is a salt formed by the combination of ranitidine and bismuth citrate. it is a new compound with unique chemical structure and physical and chemical properties, and strong hygroscopicity, light and thermal instability. |
| use | bismuth citrate ranitidine (RBC) is a salt formed by the combination of ranitidine and bismuth citrate. clinical application results show that RBC has good dual effects of anti-gastric acid secretion and protection of gastric mucosa. at the same time, combined with the use of antibiotics, it can also eradicate Helicobacter pylori infection and is an effective anti-peptic ulcer drug. |
Supplier List
CAS: 128345-62-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 128345-62-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
