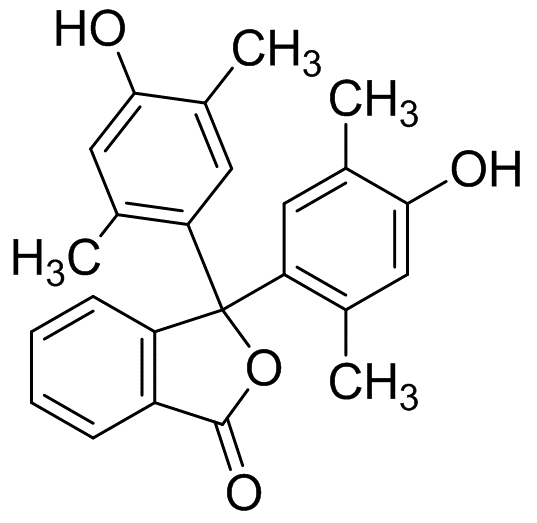
P-xylenolphthaleine
P-xylenolphthaleine
CAS: 50984-88-8
Molecular Formula: C24H22O4
P-xylenolphthaleine - Names and Identifiers
| Name | P-xylenolphthaleine |
| Synonyms | p-Xylenolphthalein P-XYLENOLPHTHALEIN P-xylenolphthaleine p-xylenolphthalein (pH 9.0-10.5) 2',5',2'',5''-TETRAMETHYLPHENOLPHTHALEIN 3,3-bis(4-hydroxy-2,5-dimethylphenyl)-2-benzofuran-1(3H)-one 3,3-Bis(4-hydroxy-2,5-dimethylphenyl)isobenzofuran-1(3H)-one 3,3-Bis(4-hydroxy-2,5-dimethylphenyl)-1(3H)-isobenzofuranone 3,3-Bis(2,5-dimethyl-4-hydroxyphenyl)isobenzofuran-1(3H)-one 4,4'-[(3-Oxo-1,3-dihydroisobenzofuran)-1,1-diyl]bis(2,5-dimethylphenol) |
| CAS | 50984-88-8 |
| EINECS | 256-893-4 |
| InChI | InChI=1/C24H22O4/c1-13-11-21(25)15(3)9-19(13)24(20-10-16(4)22(26)12-14(20)2)18-8-6-5-7-17(18)23(27)28-24/h5-12,25-26H,1-4H3 |
P-xylenolphthaleine - Physico-chemical Properties
| Molecular Formula | C24H22O4 |
| Molar Mass | 374.43 |
| Density | 1.270±0.06 g/cm3(Predicted) |
| Melting Point | 276°C |
| Boling Point | 569.6±50.0 °C(Predicted) |
| Flash Point | 198.4°C |
| Solubility | Solubility Insoluble in water; soluble in ethanol, acetone |
| Vapor Presure | 1.41E-13mmHg at 25°C |
| Appearance | powder |
| Color | Pale yellow or cream-colored |
| BRN | 346799 |
| pKa | 9.7(at 25℃) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Refractive Index | 1.649 |
| MDL | MFCD00042785 |
P-xylenolphthaleine - Risk and Safety
| WGK Germany | 3 |
P-xylenolphthaleine - Reference Information
| application | dimethylphenolphthalein can also be used as a color-changing component of children's color-changing toothpaste, and can also be used in extraction photometry for the detection of p-chloroethyl sulfur, which has important application prospects. |
| preparation | a preparation method of p-diphenolphthalein, comprising the following steps: S1, using 2 parts of p-xylene phenol and 1 part of phthalic anhydride as raw materials in molar parts, 5.5 parts of polyphosphoric acid and 0.1 parts of powdered aluminum trichloride were added for condensation reaction to obtain a mixed solution containing p-diphenolphthalein, in which the condensation reaction temperature was 70 ℃, and the condensation reaction time was 3.5h; S2, the mixed solution containing p-diphenolphthalein was cooled, and water was added for quenching reaction, in which the weight ratio of water to xylenol was 9.5:1, and the crude product was obtained by filtration; the crude and activated carbon are added to 95% ethanol for heating and reflux for 1h, then hot filtration and decolorization, cooling and crystallization, and drying to obtain p-methylphenolphthalein. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: P-XYLENOLPHTHALEIN Request for quotation
CAS: 50984-88-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 50984-88-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 3,3-Bis(4-hydroxy-2,5-dimethylphenyl)isobenzofuran-1(3H)-one Visit Supplier Webpage Request for quotationCAS: 50984-88-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: P-XYLENOLPHTHALEIN Request for quotationCAS: 50984-88-8
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Product Name: P-Xylenolphthalein Visit Supplier Webpage Request for quotation
CAS: 50984-88-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 50984-88-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: P-XYLENOLPHTHALEIN Request for quotation
CAS: 50984-88-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 50984-88-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 3,3-Bis(4-hydroxy-2,5-dimethylphenyl)isobenzofuran-1(3H)-one Visit Supplier Webpage Request for quotationCAS: 50984-88-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: P-XYLENOLPHTHALEIN Request for quotationCAS: 50984-88-8
Tel: 15335994747
Email: 3521670276@qq.com
Mobile: 15335994747
QQ: 3521670276
Wechat: A2020071098
Product Name: P-Xylenolphthalein Visit Supplier Webpage Request for quotation
CAS: 50984-88-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 50984-88-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



