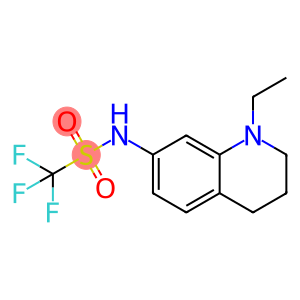
N-(1-ethyl-3,4-dihydro-2H-quinolin-7-yl)-1,1,1-trifluoro-methanesulfonamide
N-(1-ethyl-3,4-dihydro-2H-quinolin-7-yl)-1,1,1-trifluoro-methanesulfonamide
CAS: 848080-31-9
Molecular Formula: C12H15F3N2O2S
N-(1-ethyl-3,4-dihydro-2H-quinolin-7-yl)-1,1,1-trifluoro-methanesulfonamide - Names and Identifiers
N-(1-ethyl-3,4-dihydro-2H-quinolin-7-yl)-1,1,1-trifluoro-methanesulfonamide - Physico-chemical Properties
| Molecular Formula | C12H15F3N2O2S |
| Molar Mass | 308.32 |
| Density | 1.390±0.06 g/cm3(Predicted) |
| Boling Point | 371.3±52.0 °C(Predicted) |
| Flash Point | 178.337°C |
| Vapor Presure | 0mmHg at 25°C |
| pKa | 2.99±0.20(Predicted) |
| Refractive Index | 1.536 |
N-(1-ethyl-3,4-dihydro-2H-quinolin-7-yl)-1,1,1-trifluoro-methanesulfonamide - Introduction
N-(1-ethy1-3, -1, -1,-also known as N-(1-ethy1-3, -1)-1, -1,-is an organic compound.
Nature:
-Appearance: Powder
-Molecular formula: C12H16F3N2O2S
-Molecular weight: 324.33g/mol
-melting point: about 150-152 ℃
-Boiling point: about 415 ℃
-Colorless to light yellow solid
-Soluble in some organic solvents, such as dimethyl sulfoxide (DMSO) and N,N-dimethylformamide (DMF)
Use:
N-(1-ethy1-3, Ni)-1,1, Ni has many uses in the chemical field, such:
-used as an organic synthesis intermediate, involved in the construction of complex organic molecules
-Used as a coordinating reagent or catalyst to participate in organic reactions
-Can be used for high-throughput screening in drug research to find new drug molecules
Preparation Method:
N-(1-ethyl-3, Ni)-1,1, Ni has many preparation methods, one of which is a common method:
1. First, hydrofluoric acid is reacted with trifluoromethanesulfonyl chloride to obtain trifluoromethanesulfonic acid.
2. cyclization reaction with 3,4-dihydroquinoline to form dihydroquinoline under the action of base.
3. the alkylation reaction of dihydroquinoline was carried out, and ethyl bromide was added under alkali catalysis to generate 1-ethyl -3,4-dihydroquinoline.
4. Finally, trifluoromethanesulfonic acid is added to 1-ethyl-3, 4-dihydroquinoline to obtain N-(1-ethy1-3, Ni)-1,1, Ni.
Safety Information:
The specific safety information of N-(1-ethy1-3, Lin)-1,1, Lin needs to be understood through the relevant safety literature. In general, the following aspects need to be paid attention to during the use and storage of chemicals:
-Avoid direct contact with skin and eyes, and wear personal protective equipment such as gloves and safety glasses when using.
-Operate in a well-ventilated place to avoid inhaling dust or steam. In case of dyspnea and other uncomfortable symptoms, leave the operation area in time.
-Avoid contact with oxidants and strong acids to avoid chemical reactions.
-During use and storage, the relevant safety operation specifications should be observed, and the ignition point and spontaneous ignition point should be strictly controlled.
Nature:
-Appearance: Powder
-Molecular formula: C12H16F3N2O2S
-Molecular weight: 324.33g/mol
-melting point: about 150-152 ℃
-Boiling point: about 415 ℃
-Colorless to light yellow solid
-Soluble in some organic solvents, such as dimethyl sulfoxide (DMSO) and N,N-dimethylformamide (DMF)
Use:
N-(1-ethy1-3, Ni)-1,1, Ni has many uses in the chemical field, such:
-used as an organic synthesis intermediate, involved in the construction of complex organic molecules
-Used as a coordinating reagent or catalyst to participate in organic reactions
-Can be used for high-throughput screening in drug research to find new drug molecules
Preparation Method:
N-(1-ethyl-3, Ni)-1,1, Ni has many preparation methods, one of which is a common method:
1. First, hydrofluoric acid is reacted with trifluoromethanesulfonyl chloride to obtain trifluoromethanesulfonic acid.
2. cyclization reaction with 3,4-dihydroquinoline to form dihydroquinoline under the action of base.
3. the alkylation reaction of dihydroquinoline was carried out, and ethyl bromide was added under alkali catalysis to generate 1-ethyl -3,4-dihydroquinoline.
4. Finally, trifluoromethanesulfonic acid is added to 1-ethyl-3, 4-dihydroquinoline to obtain N-(1-ethy1-3, Ni)-1,1, Ni.
Safety Information:
The specific safety information of N-(1-ethy1-3, Lin)-1,1, Lin needs to be understood through the relevant safety literature. In general, the following aspects need to be paid attention to during the use and storage of chemicals:
-Avoid direct contact with skin and eyes, and wear personal protective equipment such as gloves and safety glasses when using.
-Operate in a well-ventilated place to avoid inhaling dust or steam. In case of dyspnea and other uncomfortable symptoms, leave the operation area in time.
-Avoid contact with oxidants and strong acids to avoid chemical reactions.
-During use and storage, the relevant safety operation specifications should be observed, and the ignition point and spontaneous ignition point should be strictly controlled.
Last Update:2024-04-09 21:01:54
Supplier List
Product Name: Methanesulfonamide, N-(1-ethyl-1,2,3,4-tetrahydro-7-quinolinyl)-1,1,1-trifluoro- Request for quotation
CAS: 848080-31-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 848080-31-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Methanesulfonamide, N-(1-ethyl-1,2,3,4-tetrahydro-7-quinolinyl)-1,1,1-trifluoro- Request for quotation
CAS: 848080-31-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 848080-31-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
View History
