Lx4211
Lx4211
CAS: 1018899-04-1
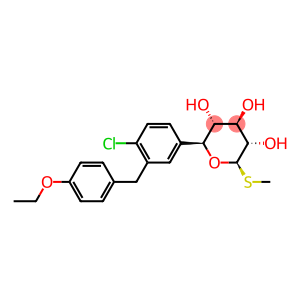
Molecular Formula: C21H25ClO5S
Lx4211 - Names and Identifiers
Lx4211 - Physico-chemical Properties
| Molecular Formula | C21H25ClO5S |
| Molar Mass | 424.94 |
| Density | 1.37±0.1 g/cm3(Predicted) |
| Boling Point | 607.9±55.0 °C(Predicted) |
| pKa | 12.87±0.70(Predicted) |
| Storage Condition | -20℃ |
| Use | LX-4211 are SGLT1/2 inhibitors and antidiabetic agents. |
| In vitro study | LX4211 inhibits [14C]AMG uptake with IC50 of 62.0 nM and 0.6 nM for mouse SGLT1 and SGLT2, respectively. |
| In vivo study | In mice, LX4211 (60 mg/kg, p.o.) decreased intestinal glucose absorption, by inhibiting SGLT1, resulted in a net increase in GLP-1 and PYY release and a decrease in GIP release and blood glucose excursion. In non-obese diabetes-prone mice with type I diabetes, Sotagliflozin (30 mg/kg) significantly improved glycemic control without increasing rates of hypoglycemia measurement. |
Lx4211 - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.353 ml | 11.766 ml | 23.533 ml |
| 5 mM | 0.471 ml | 2.353 ml | 4.707 ml |
| 10 mM | 0.235 ml | 1.177 ml | 2.353 ml |
| 5 mM | 0.047 ml | 0.235 ml | 0.471 ml |
Last Update:2024-01-02 23:10:35
Lx4211 - Reference Information
| overview | sotaglifloin LX-4211 is a dual inhibitor of SGLT1/SGLT2, which has a new hypoglycemic mechanism. existing studies have proved that it has curative effect on both type 1 diabetes and type 2 diabetes. In the three key multi-center phase III clinical studies of sotaglifloin as insulin adjuvant therapy for the treatment of type 1 diabetes, the sotaglifloin group reached the test endpoint, and no severe hypoglycemia or other SAEs occurred during the study period. This provides a clinical basis for the approval of sotaglifloin as an insulin adjuvant therapy for the treatment of type 1 diabetes. |
| application | sotaglifloin LX-4211 is a new oral diabetes drug jointly developed by Sanofi and Lescon Pharmaceuticals. As a double inhibitor of SGLT-1 and SGLT-2, the drug can reduce postprandial blood sugar, increase GLP-1 and promote urine glucose excretion. |
| mechanism of action | There are two main types of SGLT, namely SGLT1 and SGLT2. SGLT1 is mainly expressed in S3 segments of small intestinal brush border and renal proximal convoluted tubules, and a small amount is expressed in heart, trachea and brain. It is mainly responsible for transporting glucose and galactose in intestinal cavity and reabsorbing glucose that is not reabsorbed by SGLT2 in renal proximal convoluted tubules. SGLT2 is specifically expressed in the S1 segment of the proximal renal tubule, which is responsible for the reabsorption of glucose in the original urine by the proximal renal tubule, and approximately mediates the 90% renal glucose reabsorption. The mechanism of action of the listed SGLT2 inhibitors is mainly by selectively inhibiting SGLT2, reducing proximal tubular glucose reabsorption, increasing urinary glucose excretion and thus reducing blood glucose levels. As a double inhibitor of SGLT1/SGLT2, LX-4211 can not only inhibit SGLT2 and increase glucose excretion, but also inhibit SGLT1 and reduce glucose entering the blood through gastrointestinal tract, thus effectively reducing blood sugar. |
| biological activity | Sotagliflozin (LX4211, LP-802034) is an oral double SGLT1/SGLT2 inhibitor with IC50 of 36 nM and 1.8 nM respectively. Phase 3. |
| Target | Value |
| SGLT2 | 1.8 nM |
| SGLT1 | 36 nM |
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: Sotagliflozin Visit Supplier Webpage Request for quotationCAS: 1018899-04-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: LX-4211 Visit Supplier Webpage Request for quotationCAS: 1018899-04-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: LX-4211 Request for quotation
CAS: 1018899-04-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1018899-04-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: LX-4211 Visit Supplier Webpage Request for quotationCAS: 1018899-04-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Sotagliflozin Visit Supplier Webpage Request for quotationCAS: 1018899-04-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: LX-4211 Visit Supplier Webpage Request for quotationCAS: 1018899-04-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: LX-4211 Request for quotation
CAS: 1018899-04-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1018899-04-1
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: LX-4211 Visit Supplier Webpage Request for quotationCAS: 1018899-04-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



