Lixivaptan
Lixivaptan
CAS: 168079-32-1
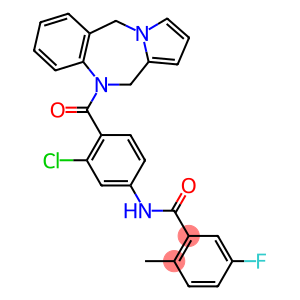
Molecular Formula: C27H21ClFN3O2
Lixivaptan - Names and Identifiers
| Name | Lixivaptan |
| Synonyms | VPA985 VPA-985 VPA 985 Lixivaptan LIXIVAPTAN WAY-VPA-985 LIXIVAPTAN (VPA 985) Lixivaptan (VPA-985) N-[3-Chloro-4-(5H-pyrrolo[2,1-c][1,4]benzodiazepin-10(11H)-ylcarbonyl)phenyl]-5-fluoro-2-methylbenzamide N-[3-chloro-4-(5H-pyrrolo[2,1-c][1,4]benzodiazepin-10(11H)-ylcarbonyl)phenyl]-5-fluoro-2-methylbenzamide N-[3-Chloro-4-(5H-pyrrolo[2,1-c][1,4]benzodiazepine-10(11H)-ylcarbonyl)phenyl]-5-fluoro-2-methylbenzamide BenzaMide,N-[3-chloro-4-(5H-pyrrolo[2,1-c][1,4]benzodiazepin-10(11H)-ylcarbonyl)phenyl]-5-fluoro-2-Methyl- N-[3-chloro-4-(6,11-dihydropyrrolo[2,1-c][1,4]benzodiazepine-5-carbonyl)phenyl]-5-fluoro-2-methyl-benzamide N-[3-Chloro-4-(10,11-dihydro-5H-pyrrolo[2,1-c][1,4]benzodiazepin-10-ylcarbonyl)phenyl]-5-fluoro-2-methylbenzamide |
| CAS | 168079-32-1 |
| InChI | InChI=1/C27H21ClFN3O2/c1-17-8-9-19(29)13-23(17)26(33)30-20-10-11-22(24(28)14-20)27(34)32-16-21-6-4-12-31(21)15-18-5-2-3-7-25(18)32/h2-14H,15-16H2,1H3,(H,30,33) |
| InChIKey | PPHTXRNHTVLQED-UHFFFAOYSA-N |
Lixivaptan - Physico-chemical Properties
| Molecular Formula | C27H21ClFN3O2 |
| Molar Mass | 473.93 |
| Density | 1.32 |
| Melting Point | >114°C (dec.) |
| Boling Point | 626.5±55.0 °C(Predicted) |
| Flash Point | 332.7°C |
| Solubility | DMSO: soluble20mg/mL, clear |
| Vapor Presure | 1.3E-15mmHg at 25°C |
| Appearance | powder |
| Color | white to beige |
| pKa | 11.86±0.70(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.657 |
| In vitro study | Lixivaptan displays competitive antagonist activity at V2 receptors. |
| In vivo study | In conscious dogs, water-loaded with 30 mL/kg (po) and arginine vasopressin (AVP)-treated (0.4 µg/kg in oil, sc), lixivaptan (1, 3, and 10 mg/kg po) increases U vol over the AVP-treated vehicle group by 438, 1018, and 1133%, respectively, while U osm decreases from 1222 mOsm/kg (water-loaded and AVP treated vehicle) to 307, 221, and 175 mOsm/kg, respectively. In homozygous Brattleboro rats lacking AVP, lixivaptan at 10 mg/kg po (i.e., 10 times the dose producing V2 antagonist activity) b.i.d. for 5 days, shows a sustained antagonist action without evidence of agonist effects. In a randomized double-blind placebo-controlled ascending single dose study, patients (deprived of fluids overnight before dosing) are dosed orally with 30, 75, or 150 mg of lixivaptan. All three doses increase urine flow and serum sodium concentrations and produced significant dose-related decreases in urinary osmolality. Phase II clinical trials in patients with congestive heart failure, liver cirrhosis with ascites or syndrome of inappropriate antidiuretic hormone have demonstrated that lixivaptan increases water clearance without affecting renal sodium excretion or activating the neurohormonal system. |
Lixivaptan - Risk and Safety
| WGK Germany | 3 |
Lixivaptan - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.11 ml | 10.55 ml | 21.1 ml |
| 5 mM | 0.422 ml | 2.11 ml | 4.22 ml |
| 10 mM | 0.211 ml | 1.055 ml | 2.11 ml |
| 5 mM | 0.042 ml | 0.211 ml | 0.422 ml |
Last Update:2024-01-02 23:10:35
Supplier List
Spot supply
Product Name: Lixivaptan Visit Supplier Webpage Request for quotationCAS: 168079-32-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: LIXIVAPTAN Request for quotation
CAS: 168079-32-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 168079-32-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: LIXIVAPTAN Visit Supplier Webpage Request for quotation
CAS: 168079-32-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 168079-32-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Lixivaptan Visit Supplier Webpage Request for quotationCAS: 168079-32-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: LIXIVAPTAN Request for quotation
CAS: 168079-32-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 168079-32-1
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: LIXIVAPTAN Visit Supplier Webpage Request for quotation
CAS: 168079-32-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 168079-32-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


