Homoharringtonine
Homoharringtonine
CAS: 26833-87-4
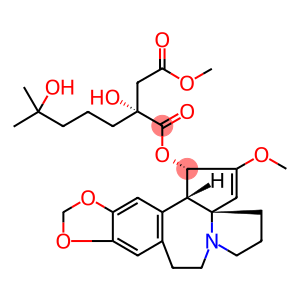
Molecular Formula: C29H39NO9
Homoharringtonine - Names and Identifiers
Homoharringtonine - Physico-chemical Properties
| Molecular Formula | C29H39NO9 |
| Molar Mass | 545.63 |
| Density | 1.2395 (rough estimate) |
| Melting Point | 144-146 C |
| Boling Point | 619.03°C (rough estimate) |
| Specific Rotation(α) | [α]/D -120 to -140°, c = 1 in chloroform-d |
| Flash Point | 385.1°C |
| Solubility | Insoluble in water, soluble in methanol, ethanol, chloroform, ether and other organic solvents. |
| Vapor Presure | 2.41E-21mmHg at 25°C |
| Appearance | White-like or yellowish crystalline powder or amorphous loose solid |
| Color | white to beige |
| pKa | 11.60±0.29(Predicted) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.6290 (estimate) |
| MDL | MFCD03410264 |
| Physical and Chemical Properties | Insoluble in water, soluble in methanol, ethanol, chloroform, ether and other organic solvents. From the genus Cephalotaxus |
| Use | Mainly used for the treatment of acute non lymphocytic leukemia |
Homoharringtonine - Risk and Safety
| Risk Codes | R26/27/28 - Very toxic by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. R28 - Very Toxic if swallowed |
| Safety Description | S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S27 - Take off immediately all contaminated clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37 - Wear suitable protective clothing and gloves. S28 - After contact with skin, wash immediately with plenty of soap-suds. |
| UN IDs | UN 1544 6.1/PG 2 |
| WGK Germany | 3 |
| RTECS | FK0260000 |
| Hazard Class | 6.1(a) |
| Packing Group | II |
Homoharringtonine - Nature
Open Data Verified Data
White crystalline or yellowish crystalline powder. Molecular weight 531. 59, melting point 71~74 ℃, bitter. Hygroscopicity, light color gradually deepened. Slightly soluble in water, soluble in methanol, ethanol, chloroform and other organic solvents.
Homoharringtonine - Preparation Method
Open Data Verified Data
The seeds, roots, stems and bark of Cephalotaxus fortunei, Cephalotaxus sinensis and Cephalotaxus sinensis were pretreated and extracted with 95% ethanol. The extract is concentrated, and the concentrated solution is transferred to the acid solution. The acid solution is extracted with chloroform treated by washing and alkalization. The total alkali was subjected to column chromatography on silica gel, and after the buffer was saturated, the total alkali was subjected to partition column chromatography, eluted with chloroform, and identified by thin layer chromatography, and the harringtonine component was collected. Chloroform was distilled off to obtain harringtonine.
Homoharringtonine - Use
Open Data Verified Data
is a non-specific drug in the cell cycle, which can inhibit the initial stage of protein synthesis in eukaryotic cells. Can be used to treat acute non-lymphatic leukemia. The color of monocytic leukemia curative effect is good, also effective for chronic myeloid leukemia.
Supplier List
CAS: 26833-87-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 26833-87-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 26833-87-4
Tel: +86-(0)731-84213302
Email: sales@staherb.cn
Mobile: +86 13875855783
QQ: 484702402
WhatsApp: +86 18374838656
Linkedin: +86 18374838656
CAS: 26833-87-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 26833-87-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 26833-87-4
Tel: +86 17733984678
Email: alice@jiuzhou-chem.com
QQ: 2284527519
WhatsApp: +86 17733984678
Linkedin: http://postmaster@api-made.com/
CAS: 26833-87-4
Tel: +86-(0)731-84213302
Email: sales@staherb.cn
Mobile: +86 13875855783
QQ: 484702402
WhatsApp: +86 18374838656
Linkedin: +86 18374838656
CAS: 26833-87-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025



