Geldanamycin
Geldanamycin
CAS: 30562-34-6
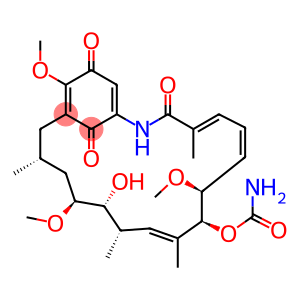
Molecular Formula: C29H40N2O9
Geldanamycin - Names and Identifiers
| Name | Geldanamycin |
| Synonyms | CS-503 U-29135 NSC122750 NSC 122750 NSC-122750 BRN 1633093 NSC 122750 GELDANAMYCIN Geldanamycin geldanamycin 30562-34-6 Geldanamycin (NSC122750) [(4Z,6E,8R,9R,10Z,12R,13R,14R,16R)-13-Hydroxy-8,14,19-trimethoxy-4,10,12,16-tetramethyl-3,20,22-trioxo-2-azabicyclo[16.3.1]docosa-4,6,10,18,21-pentaen-9-yl] carbamate (4E,6Z,8S,9S,10E,12S,13R,14S,16R)-13-hydroxy-8,14,19-trimethoxy-4,10,12,16-tetramethyl-3,20,22-trioxo-2-azabicyclo[16.3.1]docosa-1(21),4,6,10,18-pentaen-9-yl carbamate (4E,6Z,8S,9S,10E,12S,13R,14S,16R)-9-[(Aminocarbonyl)oxy]-13-hydroxy-8,14,19-trimethoxy-4,10,12,16-tetramethyl-2-azabicyclo[16.3.1]docosa-4,6,10,18,21(1)-pentene-3,20,22-trione |
| CAS | 30562-34-6 |
| EINECS | 999-999-2 |
| InChI | InChI=1/C29H40N2O9/c1-15-11-19-25(34)20(14-21(32)27(19)39-7)31-28(35)16(2)9-8-10-22(37-5)26(40-29(30)36)18(4)13-17(3)24(33)23(12-15)38-6/h8-10,13-15,17,22-24,26,33H,11-12H2,1-7H3,(H2,30,36)(H,31,35)/b10-8-,16-9+,18-13+/t15-,17+,22+,23+,24-,26+/m1/s1 |
Geldanamycin - Physico-chemical Properties
| Molecular Formula | C29H40N2O9 |
| Molar Mass | 560.64 |
| Density | 1.2443 (rough estimate) |
| Melting Point | 255 °C |
| Boling Point | 627.87°C (rough estimate) |
| Flash Point | 87℃ |
| Solubility | DMSO: soluble |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | Yellow to orange powder |
| Color | yellow to orange |
| Maximum wavelength(λmax) | ['305nm(MeOH)(lit.)'] |
| pKa | 8.42±0.70(Predicted) |
| Storage Condition | -20°C |
| Stability | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20°C for up to 3 months |
| Refractive Index | 1.5800 (estimate) |
| MDL | MFCD00274570 |
| In vitro study | Geldanamycin binds to the ATP binding site of the N-terminal domain of Hsp90s (RESIDUES 1-220). Geldanamycin dose-dependently inhibits the ATPase activity of HSP90. In the A2780 human ovarian cell line, Geldanamycin caused a dose-dependent arrest of the G2 phase and reversibly inhibited its entry into the S phase. This inhibition was accompanied by an increase in p53, which ultimately proved to be p53-dependent. Geldanamycin causes ubiquitination and proteasomal degradation of the p185 receptor protein-tyrosine kinase with an IC50 of 70 nM. |
| In vivo study | In FRE/erbB-2 mice, geldanamin (50 mg/kg) exhibited a 30% inhibition of pl85-related phosphotyrosine levels. |
Geldanamycin - Risk and Safety
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S27 - Take off immediately all contaminated clothing. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 2 |
| RTECS | LX8920000 |
| HS Code | 29419090 |
Geldanamycin - Introduction
Geldanamycin is a potent anti-tumor antibiotic. It was active in 60 cell lines at the nanomolar concentration level. It specifically binds the heat shock protein Hsp90 and its endoplasmic reticulum homolog GP96, thereby interfering with the protein's conformation and cellular stress response. In addition, it is a potent nuclear hormone receptor family inhibitor.
Last Update:2022-10-16 17:24:27
Geldanamycin - Reference Information
| properties | geldanamycin is a yellow needle-like substance, slightly soluble in water, slightly soluble in organic solvents such as methanol, ethanol, acetone, ethyl acetate, etc., soluble in chloroform, dimethyl sulfoxide, etc. The dry product is stable, and the solution state is unstable to acid, alkali, light and heat. |
| pharmacological action | geldanamycin has certain antigenic insect, anti-tumor and anti-gram positive bacteria (g) activity. GDM can inhibit the replication of various viruses in vivo, and has therapeutic effect on different models of herpes virus infection in vitro. Geldanamycin also has the effects of regulating epithelial nitrogen synthase activity and anti-inflammatory. In addition, experiments have proved that GDM and chemotherapy drugs also have synergistic effects. |
| Use | Geldanamycin (GM,GDM) is a benzoquinone-containing compound secreted by Streptomyces moist, belonging to benzoquinone Ansa (benzoquinone ansamycin) class antibiotics. In vitro experiments show that GDM has good inhibitory activity on the growth of various tumor cells and has broad-spectrum anti-proliferation and anti-tumor effects. Biochemical research |
| biological activity | Geldanamycin (NSC 122750) is a natural HSP90 inhibitor with a Kd of 1.2 μM, which specifically interferes with the combination of glucocorticoid receptor (GR)/HSP. Geldanamycin attenuates ALI (acute lung injury)/ARDS (acute respiratory distress syndrome) caused by viral infection by reducing the inflammatory response of the host. |
| Target | Value |
| p185 (SKBr3 cells) | 70 nM |
| HSP90 (N-terminal domain) (Cell-free assay) | 0.78 μM(Kd) |
| HSP90 (Cell-free assay) | 1.2 μM(Kd) |
Last Update:2024-04-09 19:05:11
Supplier List
Spot supply
Product Name: Geldanamycin Visit Supplier Webpage Request for quotationCAS: 30562-34-6
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Multiple SpecificationsSpot supply
Product Name: Geldanamycin from Streptomyces hygroscopicus Visit Supplier Webpage Request for quotationCAS: 30562-34-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Geldanamycin Request for quotation
CAS: 30562-34-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 30562-34-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: GeldanamyCln Visit Supplier Webpage Request for quotationCAS: 30562-34-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Geldanamycin Visit Supplier Webpage Request for quotationCAS: 30562-34-6
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Multiple SpecificationsSpot supply
Product Name: Geldanamycin from Streptomyces hygroscopicus Visit Supplier Webpage Request for quotationCAS: 30562-34-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Geldanamycin Request for quotation
CAS: 30562-34-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 30562-34-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: GeldanamyCln Visit Supplier Webpage Request for quotationCAS: 30562-34-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



