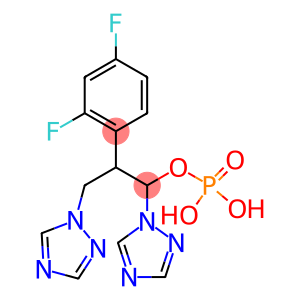
Fosfluconazole
Fosfluconazole
CAS: 194798-83-9
Molecular Formula: C13H13F2N6O4P
Fosfluconazole - Names and Identifiers
Fosfluconazole - Physico-chemical Properties
| Molecular Formula | C13H13F2N6O4P |
| Molar Mass | 386.25 |
| Density | 1.70±0.1 g/cm3(Predicted) |
| Melting Point | 223-224° |
| Boling Point | 701.5±70.0 °C(Predicted) |
| Flash Point | 378.1°C |
| Vapor Presure | 1.18E-20mmHg at 25°C |
| pKa | 1.44±0.10(Predicted) |
| Storage Condition | 2-8℃ |
| Refractive Index | 1.683 |
| In vitro study | To investigate the polarized bioconversion and the Transwell transport of phosphate prodrugs in Caco-2 monolayer, 10 μM Fosfluconazole or Fosphenytoin is dosed either in the apical or basal compartment in Transwell plates. Both prodrugs are efficiently cleaved in the apical compartment after a 2 h incubation. To further investigate the kinetics of ALP-mediated bioconversion, the concentration-dependent ALP-mediated bioconversions are conducted to determine the Michaelis-Menten constant (K m ) of prodrug bioconversion in Caco-2 monolayers. The saturation curves of Fosphenytoin and Fosfluconazole with the concentration increase are found. The estimated K m values of Fosphenytoin and Fosfluconazole are 1160 and 357 μM, respectively. |
| In vivo study | The apparent half-life for Fosfluconazole bioconversion in intestinal mucosa scraps is 10 min. Fluconazole (FLCZ) is an antifungal agent that is efficacious in the treatment of fungal peritonitis. Fosfluconazole (F-FLCZ) is the phosphate prodrug of FLCZ, which is highly soluble compared with FLCZ. F-FLCZ is useful against fungal peritonitis in continuous ambulatory peritoneal dialysis (CAPD) patients because it has a high water solubility. The aims of the present study are to characterize the peritoneal permeability of FLCZ and the pharmacokinetics of FLCZ and F-FLCZ after intraperitoneal (i.p.) administration to peritoneal dialysis rats. FLCZ or F-FLCZ is administered intravenously and intraperitoneally. After the i.p. administration of F-FLCZ, FLCZ is detected in circulating blood and the dialyzing fluid in peritoneal dialysis rats. The concentration of plasma FLCZ after the i.p. F-FLCZ administration is lower than that after the intravenous (i.v.) F-FLCZ administration. It is considered that the dose should be increased appropriately when F-FLCZ is administered intraperitoneally. The profiles of plasma FLCZ after i.v. and i.p. administrations are analyzed using a two-compartment model in which the distribution volume of the peripheral compartment is fixed at a volume of the dialyzing fluid (peritoneal dialysis PK model). The peritoneal dialysis PK model could describe the profiles of plasma and dialyzing fluid FLCZ. These results suggest that FLCZ and F-FLCZ could be administered intraperitoneally for the treatment of fungal peritonitis in CAPD patients. |
Fosfluconazole - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.589 ml | 12.945 ml | 25.89 ml |
| 5 mM | 0.518 ml | 2.589 ml | 5.178 ml |
| 10 mM | 0.259 ml | 1.294 ml | 2.589 ml |
| 5 mM | 0.052 ml | 0.259 ml | 0.518 ml |
Last Update:2024-01-02 23:10:35
Fosfluconazole - Reference Information
| biological activity | Fosfluconazole is a water-soluble fluconazole hydrochloride prodrug. Fluconazole are triazole antifungal drugs used to treat and systemic fungal infections on the tissue surface. |
| target | Antifungal |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Fosfluconazole Visit Supplier Webpage Request for quotationCAS: 194798-83-9
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Fosfluconazole Visit Supplier Webpage Request for quotationCAS: 194798-83-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Fosfluconazole Request for quotation
CAS: 194798-83-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 194798-83-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Fosfluconazole Visit Supplier Webpage Request for quotation
CAS: 194798-83-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 194798-83-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Fosfluconazole Visit Supplier Webpage Request for quotationCAS: 194798-83-9
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Fosfluconazole Visit Supplier Webpage Request for quotationCAS: 194798-83-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Fosfluconazole Request for quotation
CAS: 194798-83-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 194798-83-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Product Name: Fosfluconazole Visit Supplier Webpage Request for quotation
CAS: 194798-83-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 194798-83-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



