Epirubicin
Epirubicin
CAS: 56420-45-2
Molecular Formula: C27H29NO11
Epirubicin - Names and Identifiers
| Name | Epirubicin |
| Synonyms | WP 697 NSC 256942 Epirubicin Pidorubicin Farmarubicin Farmarubicine Pharmarubicin Epidoxorubicin 10-tetrahydro-6,8,11-trihydroxy-8-(hydroxyacetyl)-1-methoxy-(8s-)oxy)-9 |
| CAS | 56420-45-2 |
| InChI | InChI=1/C27H29NO11/c1-10-22(31)13(28)6-17(38-10)39-15-8-27(36,16(30)9-29)7-12-19(15)26(35)21-20(24(12)33)23(32)11-4-3-5-14(37-2)18(11)25(21)34/h3-5,10,13,15,17,22,29,31,33,35-36H,6-9,28H2,1-2H3/t10-,13-,15+,17-,22-,27?/m1/s1 |
Epirubicin - Physico-chemical Properties
| Molecular Formula | C27H29NO11 |
| Molar Mass | 543.52 |
| Density | 1.3783 (rough estimate) |
| Boling Point | 617.77°C (rough estimate) |
| pKa | 7.35±0.60(Predicted) |
| Storage Condition | -20°C |
| Refractive Index | 1.6400 (estimate) |
| Physical and Chemical Properties | Epirubicin Hydrochloride: C27H29 NO11? HCI. [56390-09-1]. Orange Crystal, melting point 185 C (decomposition). [Α] D 20 274 °(C = 0.01, methanol). The solution should be kept in the dark. |
| Use | For anti-tumor |
Epirubicin - Risk and Safety
| Toxicity | LD50 intravenous in dog: 2mg/kg |
Epirubicin - Reference
| Reference Show more | 1. [IF=9.867] Sun Huiying et al."CRIP1 cooperates with BRCA2 to drive the nuclear enrichment of RAD51 and to facilitate homologous repair upon DNA damage induced by chemotherapy."Oncogene. 2021 Jul;40(34):5342-5355 2. [IF=4.384] Wenxin Pei et al."High payload and targeted release of anthracyclines by aptamer-tethered DNA nanotrains — Thermodynamic and release kinetic study."Eur J Pharm Sci. 2020 May;148:105319 3. [IF=4.098] Qingying Guo et al."Spectroscopic and cytotoxicity studies on the combined interaction of (−)-epigallocatechin-3-gallate and anthracycline drugs with human serum albumin."Spectrochim Acta A. 2019 Nov;222:117213 |
Epirubicin - Reference Information
| pharmacological effects | Epirubicin (Epirubicin) is one of the clinically selected drugs for the treatment of solid tumors, interferes with and prevents the replication and transcription of DNA. It affects the function of mitochondria by inhibiting the activity of respiratory enzymes such as succinate oxidase and NADPH-oxidase. Studies have found that it can induce apoptosis of cancer cells, which may occur through the mitochondrial pathway of apoptosis. Due to its excretion via the hepatobiliary system, patients with liver dysfunction should be reduced in order to avoid accumulation of poisoning; Patients with moderate liver dysfunction (bilirubin 1.4~3mg/15% or BSP retention 9 ~), dosage should be reduced by 50%; In patients with severe hepatic impairment (bilirubin> 3mg/100ml or BSP retention> 15%), dosage should be reduced by 75%, because only a small amount of the drug is excreted by the kidneys. Usage for intravenous administration. It is recommended to inject physiological saline first to check the patency of the infusion tube and the injection needle is indeed in the vein, and then the drug is administered through the patent infusion tube. This approach reduces the risk of drug spillover and ensures intravenous saline irrigation after administration. Epirubicin injection can cause severe damage and even necrosis of the tissue, small vein injection or repeated injection of the same blood vessel will cause venous sclerosis; It can not be mixed with heparin, because the two chemical properties are not compatible, precipitation reaction occurs at a certain concentration; It can be combined with other antitumor drugs, but the dosage of epirubicin should be reduced, and the drug should not be mixed in the same syringe. Epirubicin is an anthracycline and isomer of doxorubicin. The main mechanism of action is directly embedded between DNA base pairs, interfering with the transcription process, preventing the formation of mRNA and anti-tumor effect. It can not only inhibit the synthesis of DNA, but also inhibit the synthesis of RNA, so it has an effect on all stages of the cell cycle and is a non-specific drug in the cell cycle. It also has effects on cell membranes and transport systems, but the most important site of action is the nucleus, resulting in cytotoxic effects. This product can inhibit the proliferation of mouse embryonic fibroblasts in vitro, has anti-tumor effect on animal sarcoma S180 ascites type and solid type and Gross leukemia in vivo, and is also effective for experimental L1210 and P388 breast cancer and Lewis lung cancer. |
| azithromycin | azithromycin is the first fifteen-membered macrolide antibiotic, it is also the first azalide antibiotic. Its chemical name is 9A-methyl -9-deoxy-9A-aza-9a-with erythromycin A, is derived from erythromycin A by chemical modification. It was developed by SourPliva, Yugoslavia, and was first launched in 1988 and then transferred to Pfizer (Pfi zer), USA. Its mechanism of action is similar to that of erythromycin, which acts by inhibiting the synthesis of bacterial proteins [4]. Azithromycin has strong antibacterial activity against Gram-positive bacteria and anaerobic bacteria. Compared with erythromycin, azithromycin has 4 times stronger effect on Haemophilus influenzae and Neisseria gonorrhoeae in gram-negative bacteria and 2 times stronger effect on legionella; the role of Pneumonia Mycoplasma strong 10 times; Salmonella, shigella and Escherichia coli also has a strong effect, the MIC of many pathogenic bacteria are less than 1 μg/mL, even Toxoplasma, Treponema pallidum also has a good killing effect, thus expanding the traditional effective antibacterial spectrum of macrolide drugs. At the same time, azithromycin effectively overcomes the disadvantage of easy inactivation of erythromycin in acidic environment, has high oral bioavailability, improved in Vivo Stability and pharmacokinetic properties, and has higher concentration in blood and tissue fluid, the effect is more durable, the blood Half-Life is prolonged, and the intestinal absorption is improved. Azithromycin is mainly used to treat respiratory tract infections, soft tissue infections, urinary tract infections and skin infections. Compared with other antibiotics, azithromycin has the advantages of less times of administration, shorter course of treatment and fewer side effects, therefore, it is more suitable for the treatment of mixed infection, and the clinical application is more extensive. |
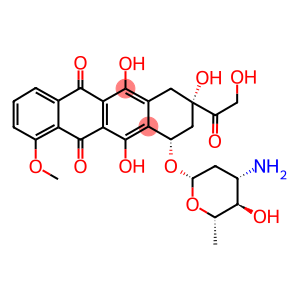
| pharmacokinetics | in the tissue distribution similar to doxorubicin, into the body rapidly distributed in the heart, kidney, liver, spleen, lung and other tissues, can not penetrate the brain blood barrier. At 1 hour, 24 hours and 48 hours after administration, the concentration in the heart and spleen was lower than that of adriamycin. The concentration in the kidney was also lower than that of adriamycin after 48 hours. The concentration in the tumor tissue was comparable to that of doxorubicin at 1 hour and 6 hours after administration and higher than that of doxorubicin at 24 hours and 28 hours after administration. In the body than Adriamycin metabolism, excretion, mainly through the liver from the biliary tract discharge. Plasma elimination half-life is 30 to 40 hours. The plasma clearance rate was about 0.9-1.14L/min. Figure 1 structural formula of epirubicin |
| uses | for breast cancer, ovarian cancer, digestive tract cancer (such as advanced gastric cancer, advanced colorectal cancer), malignant lymphoma, lung cancer and malignant melanoma. broad-spectrum anticancer antibiotics. Rapid penetration into the cell, into the nucleus, inhibit nucleic acid synthesis and mitosis. For breast cancer, malignant lymphoma, soft tissue sarcoma, gastric cancer, malignant melanoma, colorectal cancer, ovarian cancer, lung cancer, etc. for anti-tumor |
| adverse reactions | 1. Myelosuppression, mainly for white blood cells, platelets, but mild, bone marrow toxicity associated with increased dose. 2. The cardiac toxicity was lower than that of adriamycin, mainly arrhythmia and ST-T Segment Change, which could generally recover by itself. When the cumulative dose exceeds 1000mg/m2, chronic myocardial lesions may occur. Gastrointestinal reaction was Nausea, Vomit, anorexia, gastritis, Diarrhea, the incidence was lower than adriamycin. Others included alopecia, skin pigmentation, Fever, Fatigue, and dermatitis. When intravenous injection leakage can cause phlebitis, even tissue necrosis. (2016-04-14) |
| drug interaction | 1. Should avoid other drugs in the same container; 2. Can not be mixed with heparin, so as not to cause precipitation; 3. Contact with aminophylline can make the solution turn blue; 4. Can not be instilled with corticosteroids at the same time; 5. Contact with Cephalosporins can cause precipitation; 6. Can not be long-term contact with alkaline drugs or solutions; 7. With other anthracycline anticancer antibiotics can produce cross resistance; 8. With cyclophosphamide, fluorouracil, cisplatin have synergistic effect; 9. If intracavitary chemotherapy is used with cisplatin, although it has a higher efficacy, but the adverse reactions are more significant; 10. Administration of large doses of vitamin C, vitamin E can reduce myocardial toxicity; 11. Vaccination should not be carried out during medication. |
| note | . 2. Oral invalid, not intramuscular or intrathecal administration. 3. When intravenous injection, it is best to flush into the side tube after infusion to avoid extravasation or leakage of the liquid to the subcutaneous tissue, otherwise it can cause serious tissue injury and necrosis. 4. The drug can be stored at room temperature for 24 hours after dissolution, but direct sunlight should be avoided. It can be stored at 4~10 °c for 48 hours.|
| production method | compound (I) is N-acylated with trifluoroacetic anhydride to protect the amino group to obtain compound (II).. Re-oxidation to ketone (III) and then reduction to alcohol (IV). The ether bond is removed by the action of acid, and then esterified to compound (VI) by trifluoroacetic anhydride, and then subjected to selective chlorination to obtain the desired side chain (VII). The compound (VIII) is reacted with dimethyl acetone to form Ketal (IX), and then reacted with the side chain (VII) prepared above, and the obtained compound (X) is hydrolyzed, epirubicin was obtained. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 21:11:58
Supplier List
Product Name: Epirubicin Request for quotation
CAS: 56420-45-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 56420-45-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Epirubicin Visit Supplier Webpage Request for quotationCAS: 56420-45-2
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Epirubicin Visit Supplier Webpage Request for quotation
CAS: 56420-45-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 56420-45-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Epirubicin Request for quotation
CAS: 56420-45-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 56420-45-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Epirubicin Visit Supplier Webpage Request for quotationCAS: 56420-45-2
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Epirubicin Visit Supplier Webpage Request for quotation
CAS: 56420-45-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 56420-45-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


