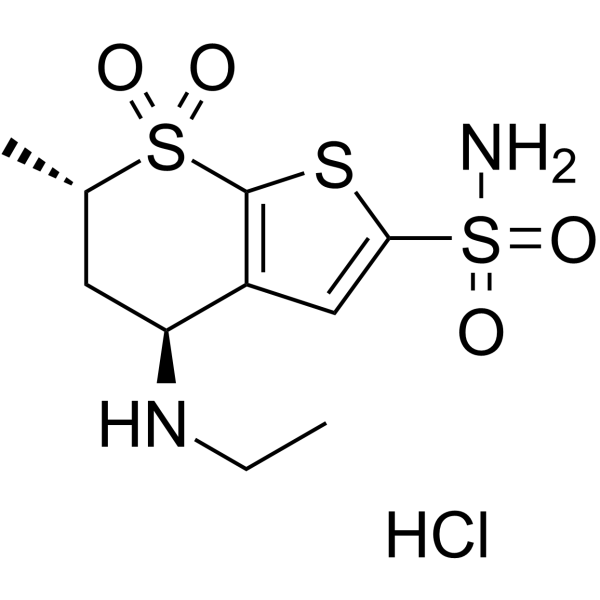
Dorzolamide.HCl
Dorzolomide hydrochloride
CAS: 130693-82-2;122028-36-8;122028-16-4;120279-36-9
Molecular Formula: C10H17ClN2O4S3
Dorzolamide.HCl - Names and Identifiers
Dorzolamide.HCl - Physico-chemical Properties
| Molecular Formula | C10H17ClN2O4S3 |
| Molar Mass | 360.9 |
| Melting Point | 265-2670C |
| Boling Point | 575.8°C at 760 mmHg |
| Specific Rotation(α) | D24 -8.34° (c = 1 in methanol) |
| Flash Point | 302°C |
| Solubility | Soluble in water (14 mg/ml at 25 °C), methanol, DMSO (<1 mg/ml at 25 °C), ethanol (s |
| Vapor Presure | 2.93E-13mmHg at 25°C |
| Appearance | White off-white crystalline powder |
| Color | White to Off-White |
| Maximum wavelength(λmax) | ['254nm(CHCl3:MeOH= aq.)(lit.)'] |
| Merck | 14,3426 |
| BRN | 5896026 |
| Storage Condition | Keep in dark place,Inert atmosphere,2-8°C |
| MDL | MFCD00884659 |
| Use | An inhibitor of carbonic anhydrase. |
| In vitro study | Dorzolamide is a powerful inhibitor of carbonic anhydrase (CA)II, which penetrates the sclera and cornea to reach the ciliary process, reducing the formation of HCO3 and aqueous humor. Dorzolamide causes retinal blood vessels to dilate blood vessels through the direct action of three different mechanisms leading to preconstriction, as well as independent changes in extracellular pH. |
| In vivo study | In glaucoma dogs, Dorzolamide, administered twice a day, three times a day, significantly reduced intraocular pressure (IOP) on the first day (7.6mm Hg twice a day, and it is 16.4 mmHg three times a day), and even higher on the fifth day (more than 10.4 mmHg twice a day and 13.9 mmHg three times a day). The combination of Dorzolamide (instillation of the drug eye for 1 to 5 days) and oral methazolamide (administration for 3 days through the 5th day) significantly reduced intraocular pressure, and this effect was on the 1st and 3rd days, and 5 days are valid. Dorzolamide(2%) was instilled three times daily to produce a similar drop in IOP compared to the combination of oral methazolamide and 2% Dorzolamide, administered twice daily. In rabbits, Dorzolamide Hydrochloride induced a very large decrease in intraocular pressure and was also effective at night. Dorzolamide maximally reduced intraocular pressure (IOP) in glaucoma monkeys by 22%,30%, and 37%, respectively. |
Dorzolamide.HCl - Risk and Safety
| WGK Germany | 3 |
| RTECS | XJ9095163 |
| HS Code | 2935904000 |
Dorzolamide.HCl - Nature
Open Data Verified Data
white to off-white crystalline powder. The melting point was 283-285 °c. Soluble in water, slightly soluble in methanol or ethanol.
Dorzolamide.HCl - Preparation Method
Open Data Verified Data
4-hydroxy 5,6-= hydro-6-tetra-4h-thieno [2,3-b] thiane-7, 7-= oxide mixed with acetonitrile, the reaction is carried out by slowly adding concentrated sulfuric acid, and the obtained product is sulfonated with chlorosulfonic acid or sulfuric acid, and then reacted with thionyl chloride to convert to sulfonyl chloride. The material in Tetrahydrofuran, the addition of ammonia, reaction to obtain sulfonamide, after reduction, that is to obtain the product, hydrochloric acid to obtain its hydrochloride.
Dorzolamide.HCl - Introduction
Dorzolamide.HCl - Use
Open Data Verified Data
developed by Merck Co, Inc., USA, was first launched in the United States in 1995. Carbonic anhydrase inhibitors. For the treatment of glaucoma.
Dorzolamide.HCl - Reference Information
| Biological activity | Dorzolamide (MK-507, L-671152) HCl is a water-soluble human carbonic anhydrase II and IV inhibitor with Ki 1.9 nM and 31 nM, respectively, used to treat glaucoma. |
| Target | Value |
| Carbonic anhydrase II | 1.9 nM(Ki) |
| Carbonic anhydrase IV | 31 nM(Ki) |
| Carbonic anhydrase I | 6000 nM(Ki) |
| Use | Function: Dozolamide (trade name Trusopt) is a carbonic anhydrase inhibitor. Dorzolamide hydrochloride is an anti-glaucoma topical eye drops. Dorzolamide hydrochloride is used to reduce the increased intraocular pressure of open-angle glaucoma and ocular hypertension |
Supplier List
CAS: 122028-36-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 130693-82-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 120279-36-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 130693-82-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 130693-82-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 122028-36-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 130693-82-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 120279-36-9
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 130693-82-2
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 130693-82-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025



