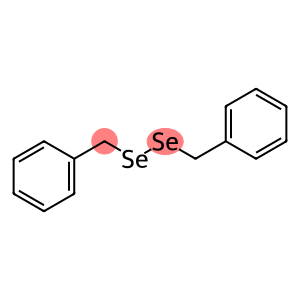
Dibenzyldiselenide
Dibenzyldiselenide
CAS: 1482-82-2
Molecular Formula: C14H14Se2
Dibenzyldiselenide - Names and Identifiers
| Name | Dibenzyldiselenide |
| Synonyms | Dibenzyldiselane BENZYL DISELENIDE Dibenzyldiselenide Dibenzyldiselenium Dibenzyl diselenide DIBENZYL DISELENIDE 1,2-Dibenzyldiselane Dibenzyl perdiselenide Bis(benzyl) perselenide Diselenide,bis(phenylmethyl) diselane, bis(phenylmethyl)- Diselenide, bis(phenylmethyl) diselane, 1,2-bis(phenylmethyl)- |
| CAS | 1482-82-2 |
| InChI | InChI=1/C14H14Se2/c1-3-7-13(8-4-1)11-15-16-12-14-9-5-2-6-10-14/h1-10H,11-12H2 |
| InChIKey | HYAVEDMFTNAZQE-UHFFFAOYSA-N |
Dibenzyldiselenide - Physico-chemical Properties
| Molecular Formula | C14H14Se2 |
| Molar Mass | 340.18 |
| Melting Point | 91-93°C(lit.) |
| Boling Point | 427.3±48.0 °C(Predicted) |
| Flash Point | 212.2°C |
| Water Solubility | Soluble in water. |
| Solubility | almost transparency in hot Toluene |
| Vapor Presure | 4.13E-07mmHg at 25°C |
| Appearance | solid |
| Color | Light yellow to Yellow to Orange |
| Exposure Limit | ACGIH: TWA 0.2 mg/m3NIOSH: IDLH 1 mg/m3; TWA 0.2 mg/m3 |
| BRN | 1877745 |
| Storage Condition | Inert atmosphere,Room Temperature |
| Sensitive | Air Sensitive |
Dibenzyldiselenide - Risk and Safety
| Risk Codes | R23/25 - Toxic by inhalation and if swallowed. R33 - Danger of cumulative effects R50/53 - Very toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S20/21 - S28 - After contact with skin, wash immediately with plenty of soap-suds. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S60 - This material and its container must be disposed of as hazardous waste. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 3283 6.1/PG 2 |
| WGK Germany | 3 |
| HS Code | 29310099 |
| Hazard Class | 6.1 |
| Packing Group | III |
Dibenzyldiselenide - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| Application | diselenide is an important organic synthesis reagent and synthesis intermediate. For example: the application of dibenzyl diselenide in the synthesis of natural products; The practical application is very wide, the application of dibenzyl diselenide in the solid phase separation of light guide materials; Can accelerate the vulcanization of nitrile rubber; in cancer research, inhibit cancer. |
| Use | inhibition of coking of RP-3 jet fuel by supercritical thermal cracking of dibenzyl diselenide. |
| preparation | at present, the conventional synthesis method of dibenzyl diselenide is the reaction of chlorinated hydrocarbon with Grignard reagent; aromatic Aldehydes directly with hydrogen selenide gas. The disadvantages of these methods are that there are many steps, complicated operation, strong alkali participating in the reaction, low yield, and direct use of hydrogen selenide gas. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Dibenzyl Diselenide Visit Supplier Webpage Request for quotationCAS: 1482-82-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Dibenzyl diselenide Request for quotation
CAS: 1482-82-2
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1482-82-2
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
Spot supply
Product Name: Dibenzyl Diselenide Visit Supplier Webpage Request for quotationCAS: 1482-82-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Dibenzyl diselenide Request for quotation
CAS: 1482-82-2
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
CAS: 1482-82-2
Tel: +8618632988332
Email: jamie@api-made.com
QQ: 2783120098
WhatsApp: +8618632988332
View History

