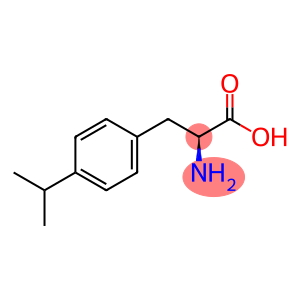
DL-p-Isopropylphenylalanine
DL-p-Isopropylphenylalanine
CAS: 98708-79-3
Molecular Formula: C12H17NO2
DL-p-Isopropylphenylalanine - Names and Identifiers
DL-p-Isopropylphenylalanine - Physico-chemical Properties
| Molecular Formula | C12H17NO2 |
| Molar Mass | 207.27 |
| Density | 1.105±0.06 g/cm3(Predicted) |
| Boling Point | 340.6±30.0 °C(Predicted) |
| pKa | 2.23±0.10(Predicted) |
DL-p-Isopropylphenylalanine - Introduction
DL-p-Isopropylphenylalanine(DL-p-Isopropylphenylalanine) is a derivative of an amino acid. The following is a description of its nature, use, preparation and safety information:
Nature:
DL-p-Isopropylphenylalanine is a colorless crystalline solid, stable at room temperature. It is a non-polar substance, hardly soluble in water, but soluble in some organic solvents such as ethanol and dimethyl sulfoxide. It is an optically active substance and has two stereoisomeric forms, D and L, because it contains two enantiomers.
Use:
DL-p-Isopropylphenylalanine has some applications in the field of medicine. It can be used as a raw material for the synthesis of certain drugs. In addition, it is also used as an antagonist, which can block specific receptors in some biological processes, and has anti-inflammatory, anti-cancer and immunomodulatory effects.
Preparation Method:
The preparation of the DL-p-Isopropylphenylalanine is generally carried out by a chemical synthesis method. The specific steps include the synthesis of phenylacetone as a starting material through esterification, carboxylic acid protection, substitution and other reactions.
Safety Information:
There is a lack of detailed information and research on the safety of DL-p-Isopropylphenylalanine. However, in general, when using and handling the substance, the safe handling of common chemicals should be observed, including wearing protective equipment and avoiding direct contact with eyes, skin and respiratory tract. In addition, the substance should be stored in a dry, cool and well-ventilated place, away from fire and oxidizing agents.
Please note that the above information is for reference only. If you need to use or handle the substance, please do it in a professional environment with relevant chemical knowledge and experimental experience.
Nature:
DL-p-Isopropylphenylalanine is a colorless crystalline solid, stable at room temperature. It is a non-polar substance, hardly soluble in water, but soluble in some organic solvents such as ethanol and dimethyl sulfoxide. It is an optically active substance and has two stereoisomeric forms, D and L, because it contains two enantiomers.
Use:
DL-p-Isopropylphenylalanine has some applications in the field of medicine. It can be used as a raw material for the synthesis of certain drugs. In addition, it is also used as an antagonist, which can block specific receptors in some biological processes, and has anti-inflammatory, anti-cancer and immunomodulatory effects.
Preparation Method:
The preparation of the DL-p-Isopropylphenylalanine is generally carried out by a chemical synthesis method. The specific steps include the synthesis of phenylacetone as a starting material through esterification, carboxylic acid protection, substitution and other reactions.
Safety Information:
There is a lack of detailed information and research on the safety of DL-p-Isopropylphenylalanine. However, in general, when using and handling the substance, the safe handling of common chemicals should be observed, including wearing protective equipment and avoiding direct contact with eyes, skin and respiratory tract. In addition, the substance should be stored in a dry, cool and well-ventilated place, away from fire and oxidizing agents.
Please note that the above information is for reference only. If you need to use or handle the substance, please do it in a professional environment with relevant chemical knowledge and experimental experience.
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: DL-p-Isopropylphenylalanine Visit Supplier Webpage Request for quotationCAS: 98708-79-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: DL-p-Isopropylphenylalanine Visit Supplier Webpage Request for quotationCAS: 98708-79-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
View History
