Celiprolol
Celiprolol
CAS: 56980-93-9
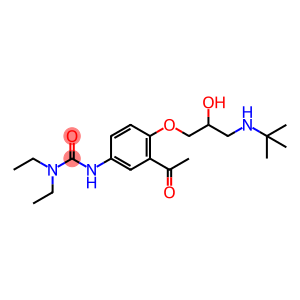
Molecular Formula: C20H33N3O4
Celiprolol - Names and Identifiers
| Name | Celiprolol |
| Synonyms | -diethyl- CELIPROLOL Celiprolol celiptolol Celiprolol Base (R,S)-Celiprolol CELIPROLOLHYDROCHLORIDE(SUBJECTTOPANTENTFREE) 3-{3-acetyl-4-[3-(tert-butylamino)-2-hydroxypropoxy]phenyl}-1,1-diethylurea 3-[3-acetyl-4-(2-hydroxy-3-tert-butylamino-propoxy)-phenyl]-1,1-diethyl-urea n'-(3-acetyl-4-(3-((1,1-dimethylethyl)amino)-2-hydroxypropoxy)phenyl)-n,n-di |
| CAS | 56980-93-9 |
| EINECS | 260-497-7 |
| InChI | InChI=1/C20H33N3O4/c1-7-23(8-2)19(26)22-15-9-10-18(17(11-15)14(3)24)27-13-16(25)12-21-20(4,5)6/h9-11,16,21,25H,7-8,12-13H2,1-6H3,(H,22,26) |
Celiprolol - Physico-chemical Properties
| Molecular Formula | C20H33N3O4 |
| Molar Mass | 379.49 |
| Density | 1.114g/cm3 |
| Melting Point | 110-112° |
| Boling Point | 586.5°C at 760 mmHg |
| Flash Point | 308.5°C |
| Water Solubility | 22.9mg/L(22.5 ºC) |
| Solubility | Chloroform (slightly), Methanol (Slightly) |
| Vapor Presure | 1.34E-14mmHg at 25°C |
| Appearance | Solid |
| Color | Yellow Sticky |
| pKa | pKa ~9.7 (Uncertain) |
| Storage Condition | -20°C Freezer, Under Inert Atmosphere |
| Refractive Index | 1.545 |
Celiprolol - Nature
Open Data Verified Data
Crystal, melting point 110~112 °c.
Celiprolol - Preparation Method
Open Data Verified Data
p-aminophenyl ether, potassium bicarbonate and acetone were mixed and reacted Dropwise with N,N-diethylchloroformamide at room temperature, followed by addition of water and adjustment to pH 6 with hydrochloric acid. After completion of the reaction, the product is obtained by washing, drying and recrystallization. The obtained compound, dichloromethane and acetyl chloride are mixed, and anhydrous aluminum trichloride is added for reaction. After the reaction, the treated compound and epichlorohydrin are mixed, A sodium hydroxide solution was added dropwise to carry out a reaction, water and excess epichlorohydrin were distilled off under reduced pressure, benzene was added, and the mixture was washed with a saturated aqueous solution of sodium chloride. The benzene was distilled off under reduced pressure, acetone was added, hydrobromic acid and water were added at room temperature and reacted again. After crystallization and drying, the obtained compound is reacted with tert-butylamine to obtain celiprolol, and the addition of hydrochloric acid to obtain celiprolol hydrochloride.
Celiprolol - Use
Open Data Verified Data
was first launched in Australia in 1985. A highly selective beta-l-receptor blocker with a vasodilator effect. This product has no membrane stabilization effect and does not inhibit myocardial contractility, and is less likely to cause sinus bradycardia than other p-receptor blockers without endogenous sympathomimetic activity. For the treatment of angina and ischemic heart disease, can reduce the frequency and extent of angina attacks, improve exercise tolerance, but to reduce resting and exercise heart rate, weaker than Atenolol.
Celiprolol - Safety
Open Data Verified Data
male mice, rats LDso (mg]kg):56.2,68.3 I. V. 1834,3826 p. O.
Celiprolol - Reference Information
| Overview | Celiolol hydrochloride (celiprololhydrochloride), the chemical name is N'-[3-acetyl-4-(3-tert-butylamino-2-hydroxypropoxy) phenyl]-N,N-diethylurea hydrochloride, developed by French Sanofi-Aventis Company, was first listed in Austria in 1983. This product has a high degree of cardiac selectivity, and has some β2-receptor excitation and weak α2-receptor blocking activity, with few and light adverse reactions. There are many reports on the synthesis process, but the starting materials and synthesis routes are mostly the same, and there is still room for improvement in terms of production costs and environmental pollution of the process. Celiprolol highly selectively binds to β1 receptors on myocardial cell membrane, and its affinity is 20-30 times stronger than β2 receptors in bronchial and vascular smooth muscle. It can reduce heart rate and cardiac output during rest and exercise, reduce systolic blood pressure during exercise, and inhibit isoproterenol-induced tachycardia. |
| drug action and clinical application | celiolol, also known as celiolol and diethylurea propanolol, is a highly selective β-receptor blocker, which dilates blood vessels and lowers blood pressure by blocking β1 receptors. It can bind to β1 receptor on myocardial cell membrane with high selectivity, and its affinity is 20-30 times stronger than β2 receptor in bronchial and vascular smooth muscle. It can reduce heart rate and cardiac output during rest and exercise, reduce systolic blood pressure during exercise, and inhibit isoproterenol-induced tachycardia. The β2 receptor-mediated vasodilation effect of isoproterenol cannot be reversed in healthy individuals. It has intrinsic sympathomimetic activity, does not increase respiratory resistance, dilates peripheral blood vessels, and improves blood circulation. There is no membrane stabilizing effect, no inhibition of myocardial contractility, and compared with other β-blockers without endogenous sympathomimetic activity, the possibility of causing sinus bradycardia is small. It is suitable for the treatment of mild to moderate hypertension. |
| pharmacokinetic study | the absorption of celiolol in vivo is dose-related. High dose can increase absorption, plasma half-life (t1/2) is about 4~5 hours, intravenous administration is about 50%, oral administration is about 10 ~ 15%, urine excretion is made within 3 days, and the rest is excreted in intestinal tract. In patients with reduced renal function, the clearance rate is reduced, the dosage should be reduced, the bioavailability of patients with liver cirrhosis is reduced, and the clearance rate is increased, and the dosage should be slightly increased. |
| usage and dosage | adults take 100~300mg morning clothes once a day. The amount of treatment varies greatly among individuals and should be gradually increased. Do not stop the drug suddenly when taking the drug for a long time. |
Supplier List
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
