Bromomethylpyridine
Bromomethylpyridine
CAS: 3430-22-6
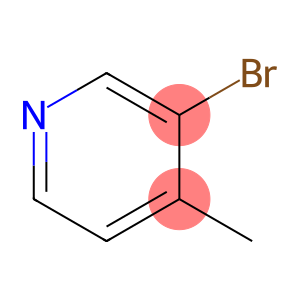
Molecular Formula: C6H6BrN
Bromomethylpyridine - Names and Identifiers
| Name | Bromomethylpyridine |
| Synonyms | 4-Meth yihebio 3-BROMO-4-PICOLINE 3-Bromo-4-picoline Bromomethylpyridine 4-METHYL-3-BROMOPYRIDINE 4-Methyl-3-Bromopyridine 3-BROMO-4-METHYLPYRIDINE 3-Bromo-4-methylpyridine |
| CAS | 3430-22-6 |
| EINECS | 608-967-6 |
| InChIKey | GSQZOLXWFQQJHJ-UHFFFAOYSA-N |
Bromomethylpyridine - Physico-chemical Properties
| Molecular Formula | C6H6BrN |
| Molar Mass | 172.02 |
| Density | 1.549g/mLat 25°C(lit.) |
| Boling Point | 199-200°C(lit.) |
| Flash Point | 175°F |
| Solubility | Chloroform, Methanol |
| Appearance | Liquid |
| Specific Gravity | 1.549 |
| Color | Clear colorless to yellow |
| BRN | 878354 |
| pKa | 3.54±0.18(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Refractive Index | n20/D 1.56(lit.) |
| Physical and Chemical Properties | Colorless transparent liquid |
Bromomethylpyridine - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S2636 - |
| UN IDs | Cool, dry,tightly closed |
| WGK Germany | 3 |
| HS Code | 29339900 |
| Hazard Class | IRRITANT |
| Packing Group | III |
Bromomethylpyridine - Reference Information
| application | 4-methyl -3-bromopyridine is an important intermediate, mainly used as pharmaceutical intermediate, organic synthesis intermediate, organic solvent, and also used in the production of dyes, fragrances and pesticides. |
| preparation | under the cooling of ice salt bath, 2-methyl -4-aminopyridine (10.8g,0.1mol) is added to 48% HBr(46ml,0.4mol), after adding, cooling to -5 ℃, slowly adding bromine (15ml,0.3mol) dropwise, and adding 30-35min, then 42g of 40% sodium nitrite solution is added dropwise below 0 ℃, the addition is completed within 1-1.1h, the addition is continued to be stirred below 0 ℃ for 30min, then 50% sodium hydroxide solution is slowly added below 20 ℃ to adjust the pH of the solution to 9, the reaction liquid is extracted with ethyl acetate, the base layer is dried with anhydrous sodium sulfate, filtered and concentrated to obtain 4-methyl -3-bromopyridine with a molar yield of 95%. |
Last Update:2024-04-09 20:52:54
Supplier List
Product Name: 3-Bromo-4-methylpyridine Request for quotation
CAS: 3430-22-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3430-22-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 3-Bromo-4-methylpyridine Visit Supplier Webpage Request for quotationCAS: 3430-22-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 3-Bromo-4-methylpyridine Visit Supplier Webpage Request for quotationCAS: 3430-22-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 3-Bromo-4-methylpyridine Request for quotation
CAS: 3430-22-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3430-22-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: 3-Bromo-4-methylpyridine Visit Supplier Webpage Request for quotationCAS: 3430-22-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: 3-Bromo-4-methylpyridine Visit Supplier Webpage Request for quotationCAS: 3430-22-6
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
View History


