Benzylphenylsulphone,
Benzylphenylsulphone,
CAS: 3112-88-7
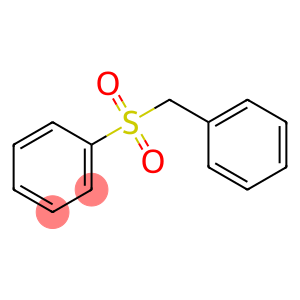
Molecular Formula: C13H12O2S
Benzylphenylsulphone, - Names and Identifiers
| Name | Benzylphenylsulphone, |
| Synonyms | (Benzylsulfonyl) Benzy Pheny Sulfone sulfone,benzylphenyl BENZYL PHENYL SULFON BENZYL PHENYL SULFONE Benzylphenylsulphone, Benzyl phenyl sulfone Benzyl phenyl sulphone BENZYL PHENYL SULPHONE (benzylsulfonyl)benzene benzenesulfonylmethylbenzene |
| CAS | 3112-88-7 |
| EINECS | 221-477-3 |
| InChI | InChI=1/C13H12O2S/c14-16(15,13-9-5-2-6-10-13)11-12-7-3-1-4-8-12/h1-10H,11H2 |
Benzylphenylsulphone, - Physico-chemical Properties
| Molecular Formula | C13H12O2S |
| Molar Mass | 232.3 |
| Density | 1.2321 (rough estimate) |
| Melting Point | 146-150 °C |
| Boling Point | 344.47°C (rough estimate) |
| Flash Point | 263.1°C |
| Vapor Presure | 6.39E-07mmHg at 25°C |
| Appearance | Crystalline Powder |
| Color | Off-white |
| BRN | 2049938 |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.5200 (estimate) |
| MDL | MFCD00025040 |
Benzylphenylsulphone, - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R22 - Harmful if swallowed |
| Safety Description | S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| RTECS | WR2700000 |
| HS Code | 29041000 |
Benzylphenylsulphone, - Reference Information
| application | benzyl phenyl sulfone can be used as an intermediate in pharmaceutical synthesis. In the research of selective oxidation of sulfide to sulfone compounds, acids, metal oxides, organic peroxides, inorganic peroxides, halogens, etc. are commonly used as oxidants. |
| preparation | 100mLSchlenk reaction tube is sequentially added with TEMPO(3.9mg,0.025mmol), methanol (1mL),CuSO(4.0mg,0.025mmol),3, 5-dimethylpyridine (28.2 μL,0.25mmol), anabl sulfide (58.8 μL,0.5mmol), and 1atm of oxygen, the sealed reaction tube is heated to 65 ℃ for 72h. After the reaction, cool to room temperature, add an appropriate amount of ethyl acetate, the blue solid in the reaction solution is precipitated, filtered, the filtrate is concentrated under reduced pressure, and purified by column chromatography to obtain the product with a yield of 82%. |
| Production method | It is obtained by the reaction of chloromethyl phenyl sulfone and phenyl gleria reagent, or benzyl phenyl sulfide is oxidized with hydrogen peroxide. |
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: BENZYL PHENYL SULFONE Request for quotation
CAS: 3112-88-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3112-88-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Benzyl Phenyl Sulfone Visit Supplier Webpage Request for quotationCAS: 3112-88-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Benzyl Phenyl Sulfone Visit Supplier Webpage Request for quotation
CAS: 3112-88-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3112-88-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: BENZYL PHENYL SULFONE Request for quotation
CAS: 3112-88-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 3112-88-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Benzyl Phenyl Sulfone Visit Supplier Webpage Request for quotationCAS: 3112-88-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Benzyl Phenyl Sulfone Visit Supplier Webpage Request for quotation
CAS: 3112-88-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 3112-88-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


