957060-88-7
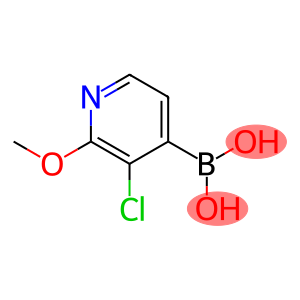
3-Chloro-2-methoxypyridine-4-boronic acid
CAS: 957060-88-7
Molecular Formula: C6H7BClNO3
957060-88-7 - Names and Identifiers
957060-88-7 - Physico-chemical Properties
| Molecular Formula | C6H7BClNO3 |
| Molar Mass | 187.39 |
| Density | 1.4g/cm3 |
| Melting Point | 140°C |
| Boling Point | 344.3°C at 760 mmHg |
| Flash Point | 162°C |
| Vapor Presure | 2.53E-05mmHg at 25°C |
| Storage Condition | 2-8°C |
| Refractive Index | 1.545 |
957060-88-7 - Introduction
Acid is an organic compound whose chemical formula is C6H7BClNO2. The following is a description of the properties, uses, preparation and safety information of the compound:
Nature:
-Appearance: acid is a white to light yellow solid.
-Solubility: Soluble in some organic solvents, such as chloroform, methanol and dimethyl sulfoxide.
-Melting point: about 152-155°C.
Use:
-must acid is an important organic synthesis intermediate, which is widely used in the synthesis of drugs, pesticides and other organic compounds. It is commonly used as a boronic acid coupling reagent in Suzuki reactions, reacting with aryl or alkynyl halides to produce the corresponding aryl or alkynyl boronic esters.
Preparation Method:
-the acid can be synthesized by the following steps:
1. 3-Chloro-2-methoxypyridine was taken as the starting material.
2. The starting material is reacted with phenylboronic acid, and the reaction is carried out in the presence of a base.
3. After the reaction and purification steps, acid is obtained.
Safety Information:
-must acid no obvious danger under normal operating conditions. However, in any laboratory operation, proper laboratory safety measures should be observed.
-Avoid contact with skin and eyes during operation, and use appropriate personal protective equipment, such as laboratory gloves, safety glasses and external laboratory ventilation.
-If you come into contact with the compound or breathe its dust, wash the affected area immediately and seek medical help.
Nature:
-Appearance: acid is a white to light yellow solid.
-Solubility: Soluble in some organic solvents, such as chloroform, methanol and dimethyl sulfoxide.
-Melting point: about 152-155°C.
Use:
-must acid is an important organic synthesis intermediate, which is widely used in the synthesis of drugs, pesticides and other organic compounds. It is commonly used as a boronic acid coupling reagent in Suzuki reactions, reacting with aryl or alkynyl halides to produce the corresponding aryl or alkynyl boronic esters.
Preparation Method:
-the acid can be synthesized by the following steps:
1. 3-Chloro-2-methoxypyridine was taken as the starting material.
2. The starting material is reacted with phenylboronic acid, and the reaction is carried out in the presence of a base.
3. After the reaction and purification steps, acid is obtained.
Safety Information:
-must acid no obvious danger under normal operating conditions. However, in any laboratory operation, proper laboratory safety measures should be observed.
-Avoid contact with skin and eyes during operation, and use appropriate personal protective equipment, such as laboratory gloves, safety glasses and external laboratory ventilation.
-If you come into contact with the compound or breathe its dust, wash the affected area immediately and seek medical help.
Last Update:2024-04-09 20:45:29
Supplier List
Spot supply
Product Name: 3-Chloro-2-methoxypyridine-4-boronic acid Visit Supplier Webpage Request for quotationCAS: 957060-88-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Chloro-2-methoxypyridin-4-ylboronic acid Request for quotation
CAS: 957060-88-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 957060-88-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 3-Chloro-2-methoxypyridine-4-boronic acid Visit Supplier Webpage Request for quotationCAS: 957060-88-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 3-Chloro-2-methoxypyridin-4-ylboronic acid Request for quotation
CAS: 957060-88-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 957060-88-7
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
View History

