936850-33-8
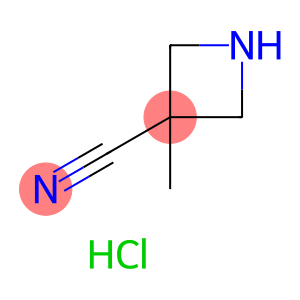
3-Cyano-3-methylazetidine hydrochloride
CAS: 936850-33-8
Molecular Formula: C5H9ClN2
936850-33-8 - Names and Identifiers
936850-33-8 - Physico-chemical Properties
| Molecular Formula | C5H9ClN2 |
| Molar Mass | 132.59 |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
936850-33-8 - Risk and Safety
| UN IDs | UN2811 |
936850-33-8 - Standard
Authoritative Data Verified Data
This strain is a mixture of phospholipids purified from chicken egg yolk or egg yolk powder by extraction with an appropriate solvent. The nitrogen (N) content should be 1.75% to 1.95% and the phosphorus (P) content should be 3 on an anhydrous basis. 5% ~ 4 .1%, containing phosphatidylcholine shall not be less than 68%, containing phosphatidylethanolamine shall not be more than 20%, containing phosphatidylcholine and phosphatidylethanolamine Total amount shall not be less than 80%.
936850-33-8 - Trait
Authoritative Data Verified Data
- This product is a milky white or light yellow powder or waxy solid, with a slight odor, and a slight smooth feeling when touched.
- This product is dissolved in ethanol, ether, chloroform or petroleum ether (boiling range 40~60°C), and is almost insoluble in acetone and water.
acid value
The acid value of this product (General 0713) shall not exceed 20.0.
saponification value
The saponification value of this product (General 0713) is 195~212.
iodine value
The iodine value of this product (General 0713) is 60~73.
perchlorination value
take this product 2.0g, precision weighing, set 250ml iodine bottle, determination according to law (General rule 0713), should not pass 3 .0.
936850-33-8 - Differential diagnosis
Authoritative Data Verified Data
- take this product O. 1g, set in the crucible, add sodium carbonate-potassium carbonate (2:1) 3g, mix, micro fire heating, the gas generated can make the wet red litmus test paper blue.
- the residue left under Item (1) of identification was about lOOmg, which was slowly burned until all the carbonates disappeared. After allowing to cool, 30ml of water was added, the residue was dissolved by slight heat and filtered, the filtrate was added to the test tube, and sulfuric acid was added dropwise until no bubbles were generated. Then 4 drops of sulfuric acid and a little potassium molybdate were added, and the mixture was heated, which should be yellow-green in color.
- in the chromatogram recorded under the item of phosphatidylcholine and phosphatidylethanolamine content determination, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the control solution.
936850-33-8 - Exam
Authoritative Data Verified Data
free fatty acids
- preparation of control solution palmitic acid was weighed. 5 1.2G, to 50ml measuring flask, dissolve and dilute to the scale with n-heptane, shake well, take 2ml accurately, to 50ml measuring flask, dilute to the scale with n-heptane, shake well, that's right.
- preparation of Test Solution take about l g of this product, weigh it accurately, put it into a 25ml measuring flask, dissolve it with isopropanol and dilute it to the scale, shake it well, then obtain it.
- measurement precision the sample solution and the reference solution were each 1 M l, placed in 20ml plug test tubes, and each was added with isopropanol-n-heptane -0. 5M o l/L sulfuric acid solution (40 : 10 : 1) mixed solution 5.0ml, shake for 1 minutes, and place for 10 minutes. Sample solution tube precision plus n-heptane 3M l and water 3ml, control solution tube precision plus n-heptane 2M l and water 4ml, plug, up and down turn 10 times, stand for at least 15 minutes, make layer. Take 3ml of the upper layer of liquid respectively, put it in a lOtnl centrifuge tube, add the indicator liquid of Nile blue (take 0.04g of Nile blue, add 20 0ml of water, dissolve it, add 100ml of n-heptane and shake it, the upper layer of n-heptane was discarded. The operation was repeated 4 times. Take 20ml of the aqueous solution of the lower layer, add 180ml of absolute ethanol, and mix. The liquid is placed in a brown bottle and can be stored at room temperature for 1 month) LML/under the condition of passing nitrogen, the solution is titrated with sodium hydroxide titration solution (O.Olmol/l) until the solution shows a pale purple color. The test solution consumed sodium hydroxide titration solution (O.Olmol/L) of the number of ML should not be greater than the control solution consumption of sodium hydroxide titration solution (O.Olmol/L) the number of milliliters (not more than 1%).
triglyceride, biliary fermentation, palmitic acid
take an appropriate amount of this product and use a mixed solvent of hexane-isopropanol-water (40 : 50: 8) to make a solution containing 20mg per lm l as a test solution, accurately weigh appropriate amounts of triglyceride reference, cholesterol and palmitic acid, respectively, and use the above mixed solvents to make triglycerides containing 0.6mg, 0.6mg and 0.2m g respectively per 1 ml, test the control solution of bile-solid fermentation and palmitic acid according to thin layer chromatography (General 0502), draw on the above test solution, triglyceride control solution and cholesterol control solution, the palmitic acid reference solution lp l was placed on the same silica gel G thin layer plate with hexane-ether-glacial acetic acid (70 : 30: 1>) as the developing solvent, the inner wall is placed in a chromatography cylinder attached with a spreading agent and moistened filter paper, and after deployment, it is taken out and dried. Spray with 1 0% (W /V) copper sulfate dilute phosphoric acid solution (8%,W /V) solution, blow dry, dry at 170°C for 10 minutes, if the test solution shows impurity spots at the corresponding position to the control solution, the color of the test solution should not be deeper than that of the control solution (I. E., triglyceride should not exceed 3%; cholesterol should not pass 2%; Palmitic acid should not pass 0 .2%).
residual solvent
take 0.2g of this product, put it in a 20ml headspace bottle, add 2ml of water, and seal it as a test solution. Accurately weigh the appropriate amount of ethanol, acetone, diethyl ether, petroleum ether and n-hexane, add water to dissolve and dilute to make the solution containing 200ug, 200ug, 200ug, 50ug, 27ug of the above solvent per lm l, respectively, as a control solution. Test as residual solvent assay (General 0861). Capillary column HP-PLOT/Q, 0.53: 30m :40um ); Initial temperature 1605°C, maintained for 8 minutes, heated to 190°C at a rate of 5°C per minute, maintained for 6 minutes; the inlet temperature was 2505°C, the detector temperature was 260°C; The split ratio was 20:1. The nitrogen flow rate was 2ml per minute. The Headspace bottle had an equilibrium temperature of 80-C, an equilibrium time of 45 minutes, and an injection volume of 1 ml. The degree of separation between chromatographic peaks shall meet the requirements. According to the external standard method to calculate the peak area, this product contains ethanol, propyl, ether shall not exceed 0.2%, containing petroleum ether shall not exceed 0.05%, containing n-hexane shall not exceed 0.02%, the total residual solvent shall not exceed 0.5%.
moisture
take this product, according to the determination of moisture (General rule 0832 first method 1), the water content shall not exceed 3%.
Heavy metals
take 2.0g of this product, slowly burn charring, add 2ml nitric acid, carefully heat to dryness, add 2ml sulfuric acid, heat to complete charring, burn to complete ashing at 500~6005°C, cold, inspection according to law (General Principles 0821 second law), containing heavy metals shall not exceed 5 parts per million.
arsenic salt
take this product l.O g, put in a Kjeldahl Flask, add 5ml of sulfuric acid, digest with small fire to carbonize (sulfuric acid can be added if necessary, the total amount is not more than 10ml), carefully add concentrated hydrogen peroxide solution Dropwise, once the reaction was stopped, heating was continued, and concentrated hydrogen peroxide solution was added dropwise until the solution was colorless. After cooling, 10ml of water was added, and the mixture was evaporated until thick smoke occurred to remove hydrogen peroxide, inspection according to law (General Principles 0822 first law), should comply with the provisions (not over 0.0002%).
microorganism
The total number of aerobic bacteria per l g of the test product shall not exceed the total number of lOOcfu, mold and yeast shall not exceed the total number of lOOcfu, the total number of fungi and yeast shall be inspected according to law (General rule 1105 and general rule 1106 ), escherichia coli shall not be detected, and salmonella shall not be detected in every 10g of the test product.
936850-33-8 - Content determination
Authoritative Data Verified Data
nitrogen
take this product about O .lg, according to the determination (General 0704).
phosphorus
- preparation of reference solution about 105g of potassium dihydrogen phosphate dried to constant weight at 0.13°C was accurately weighed, placed in a 100ml measuring flask, dissolved with water and diluted to the scale, take 10ml and put it in a 100ml measuring flask, dilute it to the scale with water, shake it, and the phosphorus (P) content in each lm l is about 30ug.
- preparation of test solution about O.lg, precision weighing, to the Crucible, add chloroform 2M l dissolved, add 2g of zinc oxide, distilled off three gas methane, slow burning to make the sample charring, then flash at 600°C for 1 hour, after cooling, l0 ml of hydrochloric acid solution (1-2) was added, the residue was dissolved by boiling for 5 minutes, transferred to a 100ml measuring flask, and diluted to the mark with water.
- determination precision: take 0, 2, 4, 6, 10ml of the reference substance, put it in 25ml measuring flask respectively, and add 10ml of water and ammonium molybdate sulfuric acid solution successively (take 5g of ammonium molybdate, add 0 .5mol/L sulfuric acid solution lOOml)lml, hydroquinone sulfuric acid solution (take hydroquinone 0.5g, add 0.25mol/L sulfuric acid solution 100ml, prepare immediately before use) lml and 50% sodium acetate solution 3ml, and diluted with water to scale, shake, and place for 5 minutes. The absorbance was measured at a wavelength of 0401 Mn by UV-Vis spectrophotometry (general rule) with the first bottle as a blank, and the regression equation was calculated with the measured absorbance and its corresponding concentration. In addition, take 4 ml of the sample solution, put it in a 25ml measuring flask, and perform the same operation according to the standard curve preparation item from "add water 10ml separately in sequence", and measure the absorbance, the phosphorus (P) content was calculated from the regression equation.
phosphatidylcholine and phosphatidylethanolamine
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system adaptability test using silica gel as a filler (column Alltima silica, 250mm X 4. 6mm X 5 pm ); Methanol-water-glacial acetic acid-triethylamine (85 :15 :0.45 :0.05) as mobile phase A, n-hexane-isopropanol-mobile phase A(20 : 48 :32) as the mobile phase B, the following table for gradient elution; Column temperature is 40C; With evaporative light scattering detector detection (reference conditions: drift tube temperature for the carrier gas flow rate of 2.0 per minute).
- Take appropriate amounts of phosphatidylethanolamine, phosphatidylinositol, lysophosphatidylethanolamine, egg yolk phosphatidylcholine, sphingomyelin and lysophosphatidylcholine, and use chloroform-methanol (2 :1) dissolve the mixed solution containing 50ug, 100ug, 100ug, 200ug, 200ug and 200ug of the above reference substance per lm l, and inject 2% 0M1 of the above solution into the human liquid chromatograph, the components should be eluted sequentially in the above order, and the separation degree of each component should be in accordance with the regulations. The number of theoretical plates should be not less than 1500 according to the peak of egg yolk phosphatidylcholine and phosphatidylethanolamine.
- determination Method: Take appropriate amount of phosphatidylethanolamine and egg yolk phosphatidylcholine reference substance respectively, weigh them precisely, and dissolve them with chloroform-methanol (2:1), diluted to prepare 6 different concentrations of phosphatidylethanolamine and phosphatidylcholine solution as a reference solution, the concentration range of phosphatidylcholine and phosphatidylethanolamine in the reference solution should cover 60% to 140% of the content of phosphatidylcholine and phosphatidylethanolamine in the test solution, precision take the above control solution 20u1 injection liquid chromatograph, with the reference solution concentration of the numerical value and the corresponding peak area of the numerical calculation of the regression equation. Another precision weigh the product about 15mg, put 50ml flask, add chloroform-methanol (2:1) dissolved and diluted to the scale, shake, as a test solution. Accurately take 20M1 injection liquid chromatograph, record chromatogram, calculate the content of phosphatidylcholine and phosphatidylethanolamine by regression equation.
936850-33-8 - Category
Authoritative Data Verified Data
pharmaceutical excipients, emulsifiers, solubilizers, etc.
936850-33-8 - Storage
Authoritative Data Verified Data
sealed, protected from light, and stored at low temperature (below 18°C).
Supplier List
CAS: 936850-33-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 936850-33-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 936850-33-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 936850-33-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 936850-33-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 936850-33-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025


