889447-19-2
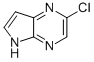
2-chloro-5H-pyrrolo[2,3-b]pyrazine
CAS: 889447-19-2
Molecular Formula: C6H4ClN3
889447-19-2 - Names and Identifiers
| Name | 2-chloro-5H-pyrrolo[2,3-b]pyrazine |
| Synonyms | LogP 2-chloro-5H-pyrrolo[2 2-Chloro-5H-pyrrolo[2,3-b... 2-Chloro-5H-pyrrolo[2,3-b]pyrazine 2-chloro-5H-pyrrolo[3,2-b]pyrazine 2-chloro-5H-pyrrolo[2,3-b]pyrazine 5H-Pyrrolo[2,3-b]pyrazine, 2-chloro- 5H-pyrrolo[2,3-b]pyrazine, 2-chloro- |
| CAS | 889447-19-2 |
| InChI | InChI=1/C6H4ClN3/c7-5-3-9-6-4(10-5)1-2-8-6/h1-3H,(H,8,9) |
889447-19-2 - Physico-chemical Properties
| Molecular Formula | C6H4ClN3 |
| Molar Mass | 153.57 |
| Density | 1.531±0.06 g/cm3(Predicted) |
| pKa | 10.63±0.50(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| Refractive Index | 1.72 |
889447-19-2 - Reference Information
| Use | 2-chloro-5h-pyrrolo [2,3-B] pyrazine is a common pyrrolo [2,3-B] pyrazine bicyclic compounds are widely used in the preparation of pharmaceutical and chemical intermediates. |
| preparation | pyrrolopyrazine bicyclic compounds and their analogs there are two synthetic strategies. Pyrazine ring is used as raw material to obtain pyrrolopyrazine bicyclic compound by nucleophilic substitution of amino alcohol, chloro and ring-closing of hydroxyl groups, the pyrrolopyrazine compound is obtained by cyclization with a keto group to form a key intermediate, reaction with ammonium acetate, and finally refluxing with phosphorus oxychloride. Preparation of 2-chloro-5h-pyrrolo [2,3-B] pyrazine starting from pyrazine-2-chloro-5-amine and ethanol, dehydration reaction preparation [1]. The synthesis reaction formula is shown in the figure below: Fig. 1 reaction formula for synthesis of 2-chloro-5h-pyrrolo [2,3-B] pyrazine N,N-dimethylformamide is added into a 250 mL three-necked bottle to start the stirring device, pyrazine-2-chloro-5-amine and anhydrous ethanol were added. Under the protection of nitrogen, the temperature was raised to 50 ° C. In the temperature-rising reactor, and the reaction was stirred for 5 hours. After completion of the reaction, the temperature was increased and left at room temperature. The reaction mixture was slowly poured into ice water, extracted with ethyl acetate (50 mL × 3), the organic phases were combined, dried over anhydrous sodium sulfate, filtered, and the filtrate was concentrated, purification by column chromatography gave a colorless oil. Add the above oily substance and anhydrous ethanol to a 250 mL three-mouth bottle, start the stirring device, add concentrated sulfuric acid Dropwise, raise the temperature to 90 ° C. In the temperature-rising reactor, and stir the reaction for 4 hours, the progress of the reaction was detected by thin layer chromatography. After completion of the reaction, the temperature was increased and left at room temperature. The reaction solution was slowly poured into ice water, extracted with ethyl acetate (50 mL × 3), combined with organic phase, dried with anhydrous sodium sulfate, filtered, the filtrate was concentrated to give 2-chloro-5h-pyrrolo [2,3-B] pyrazine as a pure product. |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2-Chloro-5H-pyrrolo[2,3-b]pyrazine Visit Supplier Webpage Request for quotationCAS: 889447-19-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-chloro-5H-pyrrolo[2,3-b]pyrazine Request for quotation
CAS: 889447-19-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 889447-19-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-chloro-5H-pyrrolo[2,3-b]pyrazine Visit Supplier Webpage Request for quotation
CAS: 889447-19-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 889447-19-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Chloro-5H-pyrrolo[2,3-b]pyrazine Visit Supplier Webpage Request for quotationCAS: 889447-19-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-chloro-5H-pyrrolo[2,3-b]pyrazine Request for quotation
CAS: 889447-19-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 889447-19-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-chloro-5H-pyrrolo[2,3-b]pyrazine Visit Supplier Webpage Request for quotation
CAS: 889447-19-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 889447-19-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


