86361-55-9
Gnetol
CAS: 86361-55-9
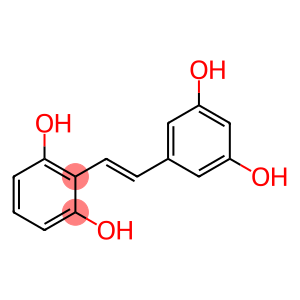
Molecular Formula: C14H12O4
86361-55-9 - Names and Identifiers
| Name | Gnetol |
| Synonyms | Gnetol -2-(3,5-Dihydroxystyryl) 2,3',5',6-Tetrahydroxy-trans-stilbene (E)-2-(3,5-Dihydroxystyryl)benzene-1,3-diol 2-[2-(3,5-dihydroxyphenyl)ethenyl]benzene-1,3-diol 2-[(1E)-2-(3,5-Dihydroxyphenyl)ethenyl]-1,3-benzenediol 1,3-Benzenediol, 2-[(1E)-2-(3,5-dihydroxyphenyl)ethenyl]- |
| CAS | 86361-55-9 |
| EINECS | 617-839-9 |
86361-55-9 - Physico-chemical Properties
| Molecular Formula | C14H12O4 |
| Molar Mass | 244.24 |
| Density | 1.468±0.06 g/cm3(Predicted) |
| Melting Point | 271°C(lit.) |
| Boling Point | 540.8±30.0 °C(Predicted) |
| Solubility | Chloroform, Dichloromethane, Ethyl Acetate, DMSO, Acetone, etc. |
| Appearance | Powder |
| Color | White to Light yellow to Light orange |
| Maximum wavelength(λmax) | ['337nm(EtOH)(lit.)'] |
| pKa | 9.06±0.40(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| MDL | MFCD12405548 |
| In vitro study | The antiproliferative activities of Gnetol are tested in HCT-116, Hep-G2, MDA-MB-231, and PC-3 cell lines by measuring cell viability after treatment with 4.1 μM, 40.9 μM, 204.7 μM, 409.4 μM, and 1023.6 μM. Gnetol shows concentration-dependent reductions in cell viability in cancer cell lines with greatest activity in colorectal cancer. Gnetol at 200 µg/mL significantly offers the highest protection of 54.3% against the toxicant. A lower dose of Gnetol (50 µg/mL) also shields the cell line from the toxic effects of CCl4. The ligand molecule TGF-β and PPARα protein show that Gnetol has the binding affinity of 7.0 and 8.4, respectively. |
| In vivo study | Male Sprague-Dawley rats were cannulated and dosed either intravenously with Gnetol (10 μg/kg) or orally (100 mg/kg). After oral and intravenous administration, Gnetol is detected in both serum and urine as the parent compound and as a glucuronidated metabolite. The bioavailability of Gnetol is determined to be 6%. Gnetol is rapidly glucuronidated and is excreted in urine and via nonrenal routes. Pretreatment of Male NIH Swiss mice (20-35 g) with Gnetol (50mg/kg, SC) is able to increase the latency period to response in analgesia models. |
86361-55-9 - Reference Information
| Introduction | 2,6,3 ',5'-tetrahydroxystilbene is also called maitenol. Buy cauline alcohol is a stilbene monomer compounds isolated from buy caulis plants, pharmacological experiments show that it has strong anti-inflammatory and antioxidant activity. |
| Application | 2,6,3 ',5'-tetrahydroxystilbene can be used to prepare 4-(6, 8-dimethoxy-2-naphthyl)-1, 3-benzenediol. 2,6,3 ',5'-tetrahydroxystilbene is a new type of 2,6,3 ',5'-tetrahydroxystilbene dimer lost two benzene ring to generate phenyl naphthalene derivatives, the results of pharmacological activity test showed that 4-(6, 8-dimethoxy-2-naphthyl)-1, 3-phenylene glycol showed strong antioxidant activity. |
| extraction method | the stem of magenge Vine was 10kg, 80% ethanol was heat extracted 3 times, the solvent was recovered under reduced pressure to obtain ethanol extract, which was dispersed with hot water and then extracted with petroleum ether, ethyl acetate and n-butanol in turn. Compounds I (20 mg), II (30 mg), III (8 mg), IV (15 mg) were separated from ethyl acetate by column chromatography and thin layer preparative chromatography, V (20 mg), VI (20 mg), VII (400 mg), VIII (100 mg), IX (33 mg), compound X (150 mg) was obtained from n-butanol fraction. IX is 2,6,3 ',5'-tetrahydroxystilbene. |
| biological activity | Gnetol is a phenolic compound isolated from the root of gneum ula bro1 N. Gnetol potently inhibited COX-1 (IC50 0.78 μm) and HDAC. Gnetol is a potent tyrosinase inhibitor with an IC50 of 4.5 μm for mouse tyrosinase, which inhibits melanin biosynthesis. Gnetol has antioxidant, antiproliferative, anticancer and hepatoprotective activities. Gnetol also has concentration-dependent α-amylase, α-glucosidase and adipogenic activity. |
Last Update:2024-04-09 15:16:53
Supplier List
Spot supply
Product Name: (E)-2-(3,5-Dihydroxystyryl)benzene-1,3-diol Visit Supplier Webpage Request for quotationCAS: 86361-55-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-[(1E)-2-(3,5-Dihydroxyphenyl)ethenyl]-1,3-benzenediol Request for quotation
CAS: 86361-55-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 86361-55-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: Gnetol Visit Supplier Webpage Request for quotationCAS: 86361-55-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (E)-2-(3,5-Dihydroxystyryl)benzene-1,3-diol Visit Supplier Webpage Request for quotationCAS: 86361-55-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-[(1E)-2-(3,5-Dihydroxyphenyl)ethenyl]-1,3-benzenediol Request for quotation
CAS: 86361-55-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 86361-55-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: Gnetol Visit Supplier Webpage Request for quotationCAS: 86361-55-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


