83643-84-9
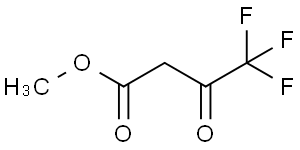
methyl trifluoroacetoacetate
CAS: 83643-84-9
Molecular Formula: C5H5F3O3
83643-84-9 - Names and Identifiers
83643-84-9 - Physico-chemical Properties
| Molecular Formula | C5H5F3O3 |
| Molar Mass | 170.09 |
| Density | 1.335g/mLat 20°C(lit.) |
| Melting Point | 19 °C |
| Boling Point | 118°C |
| Flash Point | 40°C |
| Specific Gravity | 1.340 |
| BRN | 1777343 |
| Storage Condition | Room Temprature |
| Refractive Index | n20/D 1.372 |
| MDL | MFCD00041004 |
83643-84-9 - Risk and Safety
| Risk Codes | R10 - Flammable R36/37/38 - Irritating to eyes, respiratory system and skin. R36/38 - Irritating to eyes and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S33 - Take precautionary measures against static discharges. S15 - Keep away from heat. |
| UN IDs | UN 3272 3/PG 3 |
| WGK Germany | 3 |
| HS Code | 29159000 |
| Hazard Note | Harmful/Flammable |
| Hazard Class | 3 |
| Packing Group | III |
83643-84-9 - Introduction
Methyl trifluoroacetoacetate is an organic compound with the formula C6H5F3O3. The following is an introduction to its nature, use, preparation and safety information:
Nature:
methyl trifluoroacetoacetate is a colorless liquid with a pungent odor. It is volatile at room temperature and soluble in a variety of organic solvents. It is stable to most common chemicals, but can react violently in the presence of strong oxidants.
Use:
Methyl trifluoroacetoacetate has many applications in the chemical industry. It can be used as an important intermediate in organic synthesis and can be used in the synthesis of other organic compounds, such as pesticides, pharmaceuticals and dyes. In addition, it can also be used as an organic solvent and extractant, and is widely used in chemical research and experiments.
Preparation Method:
The preparation of methyl trifluoroacetoacetate is generally obtained by reacting ethyl ketone and methyl formate under the action of a fluorinating agent (such as hydrofluoric acid or trifluoromethanesulfonic acid). The reaction is usually carried out at low temperature to improve the yield and selectivity. Detailed preparation methods can be found in handbooks of organic synthetic chemistry and in the specialized literature.
Safety Information:
Methyl trifluoroacetoacetate is a volatile organic compound, so care should be taken to avoid inhalation and skin contact during operation. It is irritating and may be harmful to the eyes, skin and respiratory system. Appropriate protective equipment such as chemical protective gloves, goggles and respiratory protective equipment must be worn when in use. In addition, it should be kept away from fire and oxidizing agents, stored in a sealed container, placed in a cool, dry place. Before use and handling, it is best to read the product safety data sheet carefully and follow the safety procedures.
Nature:
methyl trifluoroacetoacetate is a colorless liquid with a pungent odor. It is volatile at room temperature and soluble in a variety of organic solvents. It is stable to most common chemicals, but can react violently in the presence of strong oxidants.
Use:
Methyl trifluoroacetoacetate has many applications in the chemical industry. It can be used as an important intermediate in organic synthesis and can be used in the synthesis of other organic compounds, such as pesticides, pharmaceuticals and dyes. In addition, it can also be used as an organic solvent and extractant, and is widely used in chemical research and experiments.
Preparation Method:
The preparation of methyl trifluoroacetoacetate is generally obtained by reacting ethyl ketone and methyl formate under the action of a fluorinating agent (such as hydrofluoric acid or trifluoromethanesulfonic acid). The reaction is usually carried out at low temperature to improve the yield and selectivity. Detailed preparation methods can be found in handbooks of organic synthetic chemistry and in the specialized literature.
Safety Information:
Methyl trifluoroacetoacetate is a volatile organic compound, so care should be taken to avoid inhalation and skin contact during operation. It is irritating and may be harmful to the eyes, skin and respiratory system. Appropriate protective equipment such as chemical protective gloves, goggles and respiratory protective equipment must be worn when in use. In addition, it should be kept away from fire and oxidizing agents, stored in a sealed container, placed in a cool, dry place. Before use and handling, it is best to read the product safety data sheet carefully and follow the safety procedures.
Last Update:2024-04-09 21:32:43
Supplier List
Spot supply
Product Name: Methyl 4,4,4-Trifluoroacetoacetate Visit Supplier Webpage Request for quotationCAS: 83643-84-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: METHYL 4,4,4-TRIFLUOROACETOACETATE Request for quotation
CAS: 83643-84-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 83643-84-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 4,4,4-Trifluoroacetoacetic Acid Methyl Ester Visit Supplier Webpage Request for quotation
CAS: 83643-84-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 83643-84-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Methyl 4,4,4-Trifluoroacetoacetate Visit Supplier Webpage Request for quotationCAS: 83643-84-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: METHYL 4,4,4-TRIFLUOROACETOACETATE Request for quotation
CAS: 83643-84-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 83643-84-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 4,4,4-Trifluoroacetoacetic Acid Methyl Ester Visit Supplier Webpage Request for quotation
CAS: 83643-84-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 83643-84-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


