830330-21-7
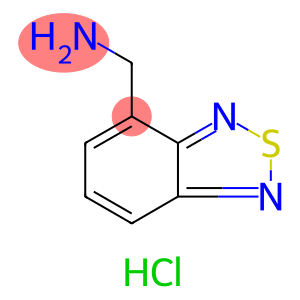
2,1,3-Benzothiadiazole-4-ylmethylamine, hydrochloride
CAS: 830330-21-7
Molecular Formula: C7H8ClN3S
830330-21-7 - Names and Identifiers
830330-21-7 - Physico-chemical Properties
| Molecular Formula | C7H8ClN3S |
| Molar Mass | 201.67 |
| Melting Point | 223-227°C |
| Boling Point | 291.9°C at 760 mmHg |
| Flash Point | 130.3°C |
| Vapor Presure | 0.00189mmHg at 25°C |
| Storage Condition | Sealed in dry,Room Temperature |
830330-21-7 - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R36 - Irritating to the eyes R22 - Harmful if swallowed |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
830330-21-7 - Introduction
2,1, hydrochloride is an organic compound with the chemical formula C8H7N3S · HCl. The following is a detailed description of its nature, use, formulation and safety information:
Nature:
-Appearance: White to off-white crystalline solid
-Melting point: about 150-155°C
-Solubility: Soluble in water and most common organic solvents
Use:
- 2,1, hydrochloride is a common intermediate in the research field of fluorescent dyes and semiconductor materials.
-It can be used in the light-emitting layer of organic light-emitting diodes (OLED), with higher luminous efficiency and lower voltage operating characteristics.
Preparation Method:
- 2,1, hydrochloride can be prepared by reacting benzothiadiazole with p-methylaniline. The reaction takes place in the presence of oxygen and the resulting product can be acidified by hydrochloric acid to give the hydrochloride form.
Safety Information:
- 2,1, hydrochloride is less toxic, but it is still necessary to follow general laboratory safety procedures.
-When using or handling this compound, care should be taken to avoid direct contact with skin and eyes. If accidental contact occurs, rinse immediately with plenty of water and get medical help.
-When storing, it should be kept in a dry, cool place, away from fire and oxidizing agents.
Nature:
-Appearance: White to off-white crystalline solid
-Melting point: about 150-155°C
-Solubility: Soluble in water and most common organic solvents
Use:
- 2,1, hydrochloride is a common intermediate in the research field of fluorescent dyes and semiconductor materials.
-It can be used in the light-emitting layer of organic light-emitting diodes (OLED), with higher luminous efficiency and lower voltage operating characteristics.
Preparation Method:
- 2,1, hydrochloride can be prepared by reacting benzothiadiazole with p-methylaniline. The reaction takes place in the presence of oxygen and the resulting product can be acidified by hydrochloric acid to give the hydrochloride form.
Safety Information:
- 2,1, hydrochloride is less toxic, but it is still necessary to follow general laboratory safety procedures.
-When using or handling this compound, care should be taken to avoid direct contact with skin and eyes. If accidental contact occurs, rinse immediately with plenty of water and get medical help.
-When storing, it should be kept in a dry, cool place, away from fire and oxidizing agents.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: 2,1,3-BENZOTHIADIAZOL-4-YLMETHYLAMINE HYDROCHLORIDE Request for quotation
CAS: 830330-21-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 830330-21-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2,1,3-BENZOTHIADIAZOL-4-YLMETHYLAMINE HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 830330-21-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 830330-21-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2,1,3-BENZOTHIADIAZOL-4-YLMETHYLAMINE HYDROCHLORIDE Request for quotation
CAS: 830330-21-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 830330-21-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2,1,3-BENZOTHIADIAZOL-4-YLMETHYLAMINE HYDROCHLORIDE Visit Supplier Webpage Request for quotation
CAS: 830330-21-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 830330-21-7
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

