82030-87-3
Somatrem
CAS: 82030-87-3
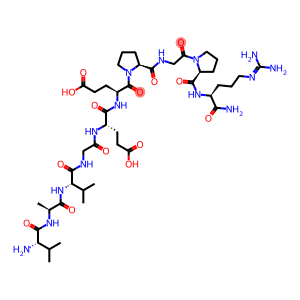
Molecular Formula: C43H72N14O14
82030-87-3 - Names and Identifiers
82030-87-3 - Physico-chemical Properties
| Molecular Formula | C43H72N14O14 |
| Molar Mass | 1009.11718 |
82030-87-3 - Nature
Open Data Verified Data
- This product is extracted from the anterior pituitary of human, cattle and sheep. Human growth hormone (HGH) is a polypeptide hormone secreted by the anterior pituitary gland and secreted by the pituitary eosinophils. It is a single polypeptide chain of 191 amino acids with two disulfides, isoelectric pH = 5.5. HGH is quite stable, and its activity can be maintained for several years under freezing conditions, and there is no change at room temperature for 48h. The length of the peptide chain of growth hormone in different species of animals is similar, but the amino acid sequence is very different, so the growth hormone of other animals has no effect on the growth of human.
- the pure product extracted from human pituitary gland was injected intramuscularly by dissolving gelatin solution, and the blood concentration reached the peak value within 2-4 hours. Mainly in the liver destruction, tl/2 only 20~25min. It stimulates the growth of almost all tissues except the brain and eyes. Hypergh in childhood leads to cytomegalovirus, and hypergh in adulthood leads to acromegaly. Young GH secretion caused by dwarfism; It can regulate metabolism, increase the cellular uptake of amino acids, promote DNA, RNA, protein synthesis, Cu, Na, Ca, P, S into the cells and bones also increased; A role of diabetes, long-term use of islet failure caused by the occurrence of diabetes.
82030-87-3 - Standard
Authoritative Data Verified Data
- This product is a protein composed of 191 amino acid residues produced by recombinant technology, and suitable excipients or stabilizers can be added. The amount of recombinant human growth hormone (C990H1528N262O300S7) per 1mg of protein should be not less than 0.91mg.
- each 1 mg of anhydrous recombinant human growth hormone corresponds to 3.0 units.
82030-87-3 - Trait
Authoritative Data Verified Data
This product is white lyophilized powder.
82030-87-3 - Use
Open Data Verified Data
early application of GH in pituitary dwarfism can significantly improve the growth rate, but does not promote precocious puberty, so it is necessary to use sex hormones at the appropriate time. Usage: intramuscular injection of 0.5 international units/kg body weight per week, divided into 2~4 times.
82030-87-3 - Differential diagnosis
Authoritative Data Verified Data
- take an appropriate amount of this product and add 0.05mol/L Tris buffer solution (adjust the pH value to 7.5 with lmol/L hydrochloric acid solution) A solution containing 2mg of recombinant human growth hormone per 1 ml was prepared by dissolving and diluting as a test solution; An appropriate amount of a recombinant human growth hormone control was additionally taken and prepared in the same manner as a control solution. According to the chromatographic condition test under the relevant protein check item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take the recombinant human growth hormone reference, add the buffer solution under identification (1) to dissolve and dilute to make a solution containing 2mg per 1 ml, take 300ul of this solution, trypsin solution [take the appropriate amount of trypsin treated by TPCK, add the buffer under identification (1) to dissolve and make a solution containing 2mg per 1 ml] 20ul and identification (1) the buffer solution under the item 30 u1, mix well, place in 37°C water bath for 4 hours, immediately place a 20°C to terminate the reaction, as a reference solution; Take this product, prepare according to the method of reference solution, as a test solution; The test solution without adding trypsin solution was taken as a blank solution. According to the high performance liquid chromatography (General 0512) test, octylsilane bonded silica gel was used as filler (5 ~ Lom); 0.1% trifluoroacetic acid solution was used as mobile phase A, the mobile phase B was 0.1% acetonitrile solution containing 90% trifluoroacetic acid; The flow rate was 1.0ml per minute; The column temperature was 35°C; The detection wavelength was 214nm. Gradient elution was performed as follows. l00ul of blank solution, reference solution and test solution were injected into the liquid chromatograph respectively, and the chromatograms were recorded, and the chromatographic peaks of the blank solution were subtracted, the peptide map of the test solution should be consistent with the peptide map of the control solution.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- take this product, add water to dissolve and dilute to make a solution containing 1 mg per 1 ml, take 90ul of this solution, add 10u1 Amphoteric electrolyte and 2ul methyl red test solution, mix well, as a test solution; Another recombinant human growth hormone control, prepared in the same method, as a control solution. Take 10 u1 of the reference solution and 10 u1 of the test solution, and add them to the upper sample hole to test by isoelectric focusing electrophoresis (General rule 0541 sixth method), the position of the main band of the test solution should be consistent with that of the reference solution.
82030-87-3 - Safety
Open Data Verified Data
local pain, swelling, fat atrophy can also occur; Occasional allergic reactions; May induce or aggravate diabetes; Long use may occur hypothyroidism, in the adenohypophysis function is reduced when especially easy to occur. Some dwarf patients lesions not in the pituitary gland, but the growth hormone releasing hormone GRH deficiency caused by, must be carefully identified. Fatal degenerative neurological disease occurred in patients treated with human pituitary GH preparations. The United States has banned the use of GH preparations derived from the human pituitary gland. GH synthesized by recombinant DNA technology is in clinical trial.
82030-87-3 - Exam
Authoritative Data Verified Data
total protein
take an appropriate amount of this product, precisely weigh it, add potassium phosphate buffer (take potassium dihydrogen phosphate 1.70g, add water 400m l to dissolve, use 0.1 mol/L sodium hydroxide solution to adjust the pH value of 7.0, diluted with water to 500ml) dissolved and quantitatively diluted to the maximum absorption wavelength (about 280nm) absorbance in 0.5~1.0 solution, as a test solution. The maximum absorption wavelength (about 280nm) and the absorbance at a wavelength of 320nm (Amax and A320) were recorded as determined by UV-Vis spectrophotometry (General 0401).
Related proteins
an appropriate amount of the product was taken, dissolved and diluted with the buffer solution under the identification (1) to prepare a solution containing 2mg of recombinant human growth hormone per 1 ml as a test solution. Butyl silane bonded silica gel as filler (5 to 10um) as determined by high performance liquid chromatography (General 0512); To identify the buffer under (1)-N-propanol (71:29) as the mobile phase, the ratio of N-propanol in the mobile phase was adjusted so that the retention time of the main peak of recombinant human growth hormone was 30-36 minutes; The flow rate was 0.5ml per minute; The column temperature was 45 ° C.; And the detection wavelength was 220nm. Take the system applicable solution [take the recombinant human growth hormone control, add the buffer under identification (1) to dissolve and dilute to make a solution containing 2mg per 1 ml, filter and sterilize, 24 hours at room temperature] 20 u1, injected into the liquid chromatograph, the separation degree between the main peak of recombinant human growth hormone and the deammoniated recombinant human growth hormone peak should be not less than 1.0, the tailing factor of the recombinant human growth hormone peak should be between 0.9 and 1.8. 20ul of the sample solution is injected into the liquid chromatograph, the chromatogram is recorded, and the total related protein shall not be more than 6.0% calculated by peak area normalization method.
high molecular protein
take an appropriate amount of this product, check according to the method under the content determination item, remove other peak areas with retention time greater than the main peak, and calculate according to the peak area normalization method, retention time less than the sum of all peak areas of the main peak shall not be greater than 4.0%.
moisture
take this product, according to the determination of moisture (General 0832 first method), the water content shall not exceed 10.0%.
sterile
take this product, dissolve it with appropriate amount of solvent, treat it by membrane filtration method, and check it according to law (General rule 1101).
bacterial endotoxin
take this product, check according to law (General rule 1143), each lmg recombinant human growth hormone containing endotoxin amount should be less than 5.0EU.
residual cell protein
take an appropriate amount of this product and check it according to law (General rule 3413). The residual amount of bacterial protein in recombinant human growth hormone per 1 mg should not exceed 10ng.
residual amount of exogenous DNA
take an appropriate amount of this product and check it according to law (General rule 3408). The host DNA in each dose of recombinant human growth hormone should not exceed 10ng.
biological activity
take this product, according to the growth hormone bioassay (General 1219) according to the law, each 1 mg of protein containing growth hormone should not be less than 2.5 units. (At least once a year)
82030-87-3 - Content determination
Authoritative Data Verified Data
measured by size exclusion chromatography (General 0514).
chromatographic conditions and system suitability test
hydrophilic modified silica gel suitable for the separation of globular proteins with a molecular weight of 5000~60000 is used as a filler; Isopropanol -0.063mol/L phosphate buffer (5.18g of disodium hydrogen phosphate anhydrous, 3.65g of sodium dihydrogen phosphate, 950ml of water was added, the pH value was adjusted to 7.0 with phosphoric acid, and 1000ml of water was made (3:97) as mobile phase; The flow rate was 0.6ml per minute; The detection wavelength was 214nm. Add 0.025mol/L phosphate buffer (pH 7.0)[take 0.063mol/L phosphate buffer (1-2.5)] dissolve and dilute to make about 1 per 1 ml. Omg solution, take 20ul injection of human liquid chromatography, recombinant human growth hormone monomer peak and dimer peak separation should meet the requirements.
assay
take this product, precision weighing, add 0.O25mol/L phosphate buffer (pH 7.0) was dissolved and quantitatively diluted to make about l per 1 ml. The solution of 0 mg is used as the test solution, and 20ul of the test solution is accurately taken and injected into the human liquid chromatograph, and the chromatogram is recorded. Another recombinant human growth hormone reference substance is taken and determined by the same method. According to the external standard method to calculate the peak area, that is.
82030-87-3 - Category
Authoritative Data Verified Data
growth hormone drugs.
82030-87-3 - Storage
Authoritative Data Verified Data
sealed and stored at 2-8°C.
82030-87-3 - Legal requirements
Authoritative Data Verified Data
This product is a recombinant DNA technology products, the production process should meet the "human recombinant DNA technology products in general" (three general) requirements.
82030-87-3 - Saizen (Somatropin for Injection)
Authoritative Data Verified Data
This product is a sterile lyophilized product of recombinant human growth hormone. The content of recombinant human growth hormone (C990H1528N262O300S7) should be 90.0% to 110.0% of the scalar quantity.
trait
This product is white lyophilized powder.
identification
This product was taken, and the same results were shown according to the tests of (1) and (4) under the item of recombinant human growth hormone.
examination
- the pH value of this product is dissolved in water and diluted to prepare a solution containing 0631 mg per lml, which is determined according to law (General Rule 6.5). The pH value should be 8.5.
- clarity and color of solution take this product, add water to dissolve and dilute to make a solution containing 0901 mg per 1 ml, and check according to law (General rules 0902 1 and 1), the solution should be clear and colorless; If it is turbid, it should not be more concentrated than the No. 2 Turbidity standard solution.
- the relevant protein should be taken and checked according to the method of recombinant human growth hormone, and the relevant protein should not be more than 13.0%.
- high molecular protein take this product, according to the method of recombinant human growth hormone, high molecular protein should not exceed 6.0%.
- the moisture content of this product shall not exceed 0832 as determined by the method for determination of moisture (General rule 3.0% first method).
- visible foreign body take this product, after each bottle plus water for injection is dissolved, check according to law (General rule 0904), shall not detect metal debris, glass debris, visible foreign bodies with a length or maximum particle size exceeding 2mm, such as cilia and clumps.
- abnormal toxicity: take this product, add sodium chloride injection to dissolve and make it into lml containing recombinant human growth hormone 1.6mg, and check it according to law (General rule 1141), the requirements shall be met.
- bacterial endotoxin and sterile take this product, according to recombinant human growth hormone under the method of inspection, should comply with the provisions.
- others should comply with the relevant provisions under injection (General 0102).
Content determination
take 5 bottles of this product, add 0. O25mol/L phosphate buffer (pH 7.0) appropriate amount, so that the content is dissolved, 5 bottles of the total amount of mixing, shake and quantitative dilution to make about 1 per 1 ml. 0 mg of the solution was used as a test solution. According to the method under the item of recombinant human growth hormone, obtained.
category
Same as recombinant human growth hormone.
specification
(1 ) 0.8mg (2)0.85mg (3)1.Omg(4)1.2mg (5)1.33mg (6)1.6mg (7)1.7mg (8)2.Omg(9)3.7mg (10)4.Omg
storage
light shielding, closed, 2~8°C storage.
82030-87-3 - Recombinant Human Growth Hormone Solution
Authoritative Data Verified Data
This product is produced by recombinant technology consisting of 191 amino acid residues of protein 9 quality solution, can add the right amount of excipients or stabilizers. The amount of recombinant human growth hormone (C990H1528N262O300S7) per 1 mg of protein should be not less than 0.9 1 mg.
System Requirements
This product is a recombinant DNA technology products, the production process should meet the "human recombinant DNA technology products in general" (three general) requirements.
trait
This product is colorless clear or slightly turbid liquid.
identification
take the product., according to the recombinant human growth hormone under the identification test, showed the same results.
examination
- total protein precision take appropriate amount of this product, use phosphate buffer (take potassium dihydrogen phosphate 1.70g, add water 400ml to dissolve, use 0.1 mol/L sodium hydroxide adjust pH to 7.0, diluted with water to 0.5 ml) quantitative dilution made at the maximum absorption wavelength (about 280nm) absorbance in 1.0 ~ solution, as a test solution, record the absorbance at the maximum absorption wavelength (about 280nm) and 320nm wavelength (Amax and A320) as determined by UV-Vis spectrophotometry (General rule 0401), the content of total protein in the test solution was calculated as mg by the following formula.
- related protein, macromolecular protein, residual body protein, residual exogenous DNA, bacterial endotoxin and biological activity, all shall be in accordance with the provisions.
- sterile take this product, by membrane filtration method, inspection according to law (General 1101 ), should comply with the provisions.
Content determination
an appropriate amount of the product was quantitatively diluted with 0.025mol/L phosphate buffer (pH 7.0) to prepare a solution containing 1.0 mg per 1 ml as a test solution. Determined according to the method under recombinant human growth hormone.
category
Same as recombinant human growth hormone.
storage
sealed, a 20°C storage.
82030-87-3 - Reference Information
| Definition | Recombinant human growth hormone (recombinant human growth, referred to as r-hGH) is produced by recombinant DNA technology. Its chemical 2 structure includes amino acid sequence and amino acid composition. It has been identified as being exactly the same as pituitary growth hormone. Recombinant human growth hormone (r-hGH), whose molecular formula is C990H1529N2630299S7, is a polypeptide chain composed of 191 amino acids. It is connected to 165 at position 53 and between 182 and 189 by disulfide bonds, and the relative molecular mass is 22125. |
| treatment of dwarfism | recombinant human growth hormone (rhGH) was launched in 1985. years of clinical research and medication experience show that it has a very definite effect on dwarfism. From its initial use in the treatment of growth hormone deficiency, to now it has been approved for the treatment of short stature caused by some other causes (such as congenital ovarian hypoplasia, small for gestational age, Prader-Willi syndrome, chronic renal failure, idiopathic short stature, etc.), rhGH has gradually become the main clinical drug for the treatment of short stature in children. |
| pharmacological action | promote height development and neural development: rhGH can stimulate target organs such as liver to produce insulin-like growth factor 1(IGF-1), which can promote bone and articular cartilage and epiphyseal chondrocytes proliferate, accelerate the linear growth of bones, and thus increase the body. At the same time, rhGH can also induce vitamin D activation, it has a synergistic effect with sex hormones and calcification hormones, promotes the absorption of calcium by bones [2], and further promotes height growth. In addition, studies have found that growth hormone and IGF-1 play an important role in human cognitive functions such as learning and memory [3]. Early application of rhGH therapy can also avoid cognitive impairment during the critical period of brain development in children with growth hormone deficiency. Regulating body substance metabolism: rhGH has a significant effect on protein and fat metabolism. On the one hand, it can promote the transport of amino acids into cells and accelerate protein synthesis; on the other hand, it can also directly act on fat cells to promote fat degradation and metabolism, thereby effectively reducing body fat and increasing muscle ratio [4]. rhGH also has the functions of enhancing myocardial function, improving immune function, increasing endometrial thickness, promoting oocyte maturation and improving sperm quality [5], so it has certain clinical significance in adult GHD, burn, assisted reproduction and other fields. |
| dosage form | currently, rhGH has three dosage forms: short-acting freeze-dried powder injection, short-acting water injection and long-acting water injection. In fact, in terms of the effectiveness and safety of drugs, there is no significant difference between the three, mainly because there are certain differences in use and price. Among them, the price of short-acting freeze-dried powder injection is relatively low. It is made by freeze-drying technology. Due to its low water content, it is easy to maintain long-term stability. However, the disadvantage is that it needs to be added with water for injection to dissolve again to restore it to water. Therefore, it is relatively troublesome to operate. When the water injection is made, the protein liquid stabilization technology is used, which avoids the freeze-drying process, does not need to be redissolved when used, and the operation is simple and can be directly injected. In addition, short-acting freeze-dried powder injection and short-acting water injection need to be injected every day, which may cause missed beating due to children's inability to cooperate for a long time or busy family members, thus affecting the therapeutic effect. The long-acting water injection only needs to be injected once a week, so that children no longer need daily injections, thus greatly improving the treatment compliance of short children. At present, rhGH has three dosage forms: short-acting freeze-dried powder injection, short-acting water injection and long-acting water injection. In fact, in terms of the effectiveness and safety of drugs, there is no significant difference between the three, mainly because there are certain differences in use and price. Among them, the price of short-acting freeze-dried powder injection is relatively low. It is made by freeze-drying technology. Due to its low water content, it is easy to maintain long-term stability. However, the disadvantage is that it needs to be added with water for injection to dissolve again to restore it to water. Therefore, it is relatively troublesome to operate. When the water injection is made, the protein liquid stabilization technology is used, which avoids the freeze-drying process, does not need to be redissolved when used, and the operation is simple and can be directly injected. In addition, short-acting freeze-dried powder injection and short-acting water injection need to be injected every day, which may cause missed beating due to children's inability to cooperate for a long time or busy family members, thus affecting the therapeutic effect. The long-acting water injection only needs to be injected once a week, so that children no longer need daily injections, thus greatly improving the treatment compliance of short children. At present, rhGH has three dosage forms: short-acting freeze-dried powder injection, short-acting water injection and long-acting water injection. In fact, in terms of the effectiveness and safety of drugs, there is no significant difference between the three, mainly because there are certain differences in use and price. Among them, the price of short-acting freeze-dried powder injection is relatively low. It is made by freeze-drying technology. Due to its low water content, it is easy to maintain long-term stability. However, the disadvantage is that it needs to be added with water for injection to dissolve again to restore it to water. Therefore, it is relatively troublesome to operate. When the water injection is made, the protein liquid stabilization technology is used, which avoids the freeze-drying process, does not need to be redissolved when used, and the operation is simple and can be directly injected. In addition, short-acting freeze-dried powder injection and short-acting water injection need to be injected every day, which may cause missed beating due to children's inability to cooperate for a long time or busy family members, thus affecting the therapeutic effect. The long-acting water injection only needs to be injected once a week, so that children no longer need daily injections, thus greatly improving the treatment compliance of short children. |
Supplier List
CAS: 82030-87-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 82030-87-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
