81334-34-1
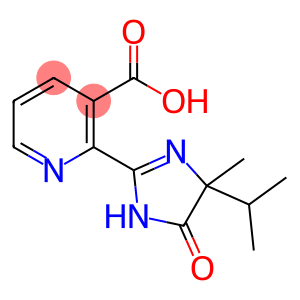
Imazapyr acid
CAS: 81334-34-1
Molecular Formula: C13H15N3O3
81334-34-1 - Names and Identifiers
81334-34-1 - Physico-chemical Properties
| Molecular Formula | C13H15N3O3 |
| Molar Mass | 261.28 |
| Density | 1.1923 (rough estimate) |
| Melting Point | 169-173°C |
| Boling Point | 404.53°C (rough estimate) |
| Flash Point | 224°C |
| Vapor Presure | 0-0Pa at 20-25℃ |
| Appearance | grayish white crystal |
| BRN | 5442754 |
| pKa | 1.9, 3.6(at 25℃) |
| Storage Condition | 0-6°C |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Sensitive | It is corrosive and cannot be mixed and stored in unlined containers. It reacts with acid, alkali an |
| Refractive Index | 1.5600 (estimate) |
| MDL | MFCD00144470 |
| Physical and Chemical Properties | The appearance was a white solid. m. P. 169-173 °c. 45 C can be stable for 3 months, room temperature can be stable for 2 years. In the pH value of 5~9, dark, stable in water medium. The half-life of hydrolysis was 6d (pH 5~9) in sunlight, and the Half-Life was 3~4 months in soil. It is corrosive and cannot be mixed and stored in an un-lined container. Reaction with acid, alkali and strong oxidant. isopropylamine salt is a white solid. 128 to 130 °c, vapor pressure 0.013 x 10-3Pa (60 °c). 15 ° C solubility: dimethylformamide 473g/L, dimethyl sulfoxide 665G/L, methanol 230g/L, ethanol 72g/L, dichloromethane 72g/L, acetone 6g/L, toluene 3G/L, water 9.74g/L, water (25 °c) 11.3g/L. |
| Use | It is a new type of broad-spectrum herbicide, which has excellent herbicidal activity on the weeds of Cyperaceae, annual and perennial monocotyledonous weeds and broad-leaved weeds. Its mechanism of action is to inhibit the synthesis of branched chain amino acids. This product is a selective herbicide, used in railway, highway, factory, warehouse, canal and forestry herbicidal, dosage 500 ~ 2000g/hm2, can control most annual and perennial herbs and plants, soil treatment and post-emergence stem and leaf treatment can be carried out, and post-treatment of seedlings is more effective. In the treatment of stems and leaves, 0.25% Non-ionic surfactant should be added to the solution. After 2 to 4 weeks, the herbs lose their green and their tissues become bad, the young leaves of the tree turned red or brown within 1 month, and some tree species were all fallen within 3 months, and eventually died. |
81334-34-1 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | R36 - Irritating to the eyes R52/53 - Harmful to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| WGK Germany | 2 |
| RTECS | US5682500 |
| HS Code | 29333990 |
| Toxicity | LD50 orally in rats: >5000 mg/kg; dermally in rabbits: >2000 mg/kg (Paxman) |
81334-34-1 - Reference Information
| LogP | -3.97-0.04 at 20℃ and pH3-9.9 |
| dissociation constant | 1.7-11.1 at 20 ℃ |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| organic heterocyclic herbicide | , it is a kind of herbicide, and its isopropylamine salt can be used before or after Bud, and can be quickly absorbed by plant roots and leaves, inhibit the biosynthesis of plant side chain amino acids, and prevent the growth of weeds, contributed to his death. The control targets include all weeds, and have good herbicidal activity against Cyperaceae weeds, annual and perennial monocotyledonous weeds, broadleaved weeds and miscellaneous trees. Applicable to non-cultivated land and rubber plantation, oil palm, forest and tea garden.|
| toxicity | Acute oral LD50>5000mg/kg, acute oral LD50>2000mg/kg in mice and acute percutaneous LD50>2000mg/kg in rabbits. It has moderate irritation to rabbit skin and irritation to eyes, but it can recover. Rainbow trout, blue gill fish LC50>100mg/L(96H), Daphnia LC50>100mg/L (48h). Quail and wild duck acute oral LD50>2150mg/kg,LC50>5000mg/kg feed (8d). Bee exposure LD50>0.1mg/only. This product isopropylamine salt of rat acute oral LD50>10000mg/kg, mice acute oral LD50>10000mg/kg, mice acute percutaneous LD50>2000mg/kg. |
| Use | Imidazolinone herbicides. Its mechanism of action is to inhibit the synthesis of branched chain amino acids. This product is a selective herbicide, used in railway, highway, factory, warehouse, canal and forestry herbicidal, dosage 500 ~ 2000g/hm2, can control most annual and perennial herbs and plants, soil treatment and post-emergence stem and leaf treatment can be carried out, and post-treatment of seedlings is more effective. In the treatment of stems and leaves, 0.25% Non-ionic surfactant should be added to the solution. After 2 to 4 weeks, the herbs lose their green and their tissues become bad, the young leaves of the tree turned red or brown within 1 month, and some tree species were all fallen within 3 months, and eventually died. It is a new broad-spectrum herbicide, which has excellent herbicidal activity on the weeds of Cyperaceae, annual and perennial monocotyledonous weeds and broadleaf weeds. Its mechanism of action is to inhibit the synthesis of branched chain amino acids. This product is a selective herbicide, used in railway, highway, factory, warehouse, canal and forestry herbicidal, dosage 500 ~ 2000g/hm2, can control most annual and perennial herbs and plants, soil treatment and post-emergence stem and leaf treatment can be carried out, and post-treatment of seedlings is more effective. In the treatment of stems and leaves, 0.25% Non-ionic surfactant should be added to the solution. After 2 to 4 weeks, the herbs lose their green and their tissues become bad, The young leaves of the tree turned red or brown within 1 month, and some tree species all fell within 3 months, and eventually died.; |
| production method | preparation method diethyl butanone diacid was synthesized by adding 48.0 ml of diethyl ether and g of sodium ethoxide. Cool to 4-6 °c. A mixture of 68.8G of diethyl oxalate and 49.7G of ethyl acetate was added dropwise. Stir for 0.5h. The temperature was increased to 38-40 °c. Reflux for 1H. Left overnight. Adjust the pH value to 1~2 with 10% dilute sulfuric acid. The ether layer was separated. Wash with water. The ether layer was distilled off, and the residue was distilled under reduced pressure to collect 130g of a 132 ° C. To 39.3 ° C./3200Pa fraction. Synthesis of diethyl 2-aminosuccinate 89.5G of diethyl butanedioate and 150g of toluene were added. The mixture was heated and stirred to 100-110 °c. Ammonia gas was then introduced for 8H. Cooling. The reaction solution was washed with water. Toluene was removed under reduced pressure. By distillation under reduced pressure, 110g of a 114-70.15 °c/666.7Pa fraction was collected. Synthesis of diethyl 5-ethylpyridine-2, 3-carboxylate 2-ethylacrolein (30.58g), diethyl 2-aminosuccinate (59.18g), p-toluenesulfonic acid (2.7g) and g of DMF were added. The reaction was carried out for 5h at 90 °c. DMF was distilled off under reduced pressure. 500g of toluene was added to the residue. Wash with 5% sodium hydroxide. The toluene was distilled off under reduced pressure. The residue was distilled under reduced pressure. The 148-150 °c/266.6Pa fraction, 70.5g, was collected. Synthesis of 5-ethylpyridine-2, 3-carboxylic acid anhydride 25% g of sodium hydroxide solution was added. The mixture was heated to 55 °c with stirring. 56.2g of diethyl 5-ethylpyridine-2, 3-carboxylate was added dropwise. The addition time was about 20min. It was heated to 65 °c for 15min. Ethanol-water was distilled off under reduced pressure. 70g of water and 318g of tetrahydrofuran were then added. The temperature was maintained at 40 °c. The reaction solution was adjusted to pH 50% with 1.65 sulfuric acid. The aqueous phase was separated. The oil phase was distilled off tetrahydrofuran. Acetic anhydride 50.4g and 4-picoline 19.1g were added. Stir at room temperature for 1H. Low boiling was distilled off under reduced pressure. 36.0G (content 81.52%) of 5-ethylpyridine-2, 3-carboxylic anhydride was obtained. Imidazole nicotinic acid synthesis 7.31G of 5-ethylpyridine-2, 3-carboxylic anhydride and 5.2g of 2-amino -2, 3-dimethylbutanamide were added along with 100ml of toluene. Stir overnight at room temperature. 2.02g of triethylamine (0.02mol) was added. Reflux dehydration. The toluene was removed under reduced pressure. The residue was added to 100ml of an aqueous solution. The pH was adjusted to 9 with 10% sodium hydroxide. The oil phase was separated. The aqueous phase was adjusted to PH 3 with hydrochloric acid. The precipitated solid was recrystallized from a mixed solvent. The product was obtained as a colorless solid (7.3g). m. P. 168-170 °c. Preparation Method: dipyridinedicarboxylic acid loses water to generate pyridine dianhydride, then reacts with (CH3)2CHCCH3(CN)NH2 to generate carbamoyl nicotinic acid, and then hydrolyzes with hydrogen peroxide in an aqueous solution of sodium hydroxide to warm and cyclize, synthesis of imidazole nicotinic acid. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:41:03
Supplier List
Spot supply
Product Name: Imazamox Impurity 21 Visit Supplier Webpage Request for quotationCAS: 81334-34-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Imazapyr Visit Supplier Webpage Request for quotationCAS: 81334-34-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Imazapyr acid Request for quotation
CAS: 81334-34-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 81334-34-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Imazapyr acid Request for quotation
CAS: 81334-34-1
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 81334-34-1
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Multiple SpecificationsSpot supply
Product Name: Imazapyr Acid Visit Supplier Webpage Request for quotationCAS: 81334-34-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Imazamox Impurity 21 Visit Supplier Webpage Request for quotationCAS: 81334-34-1
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: Imazapyr Visit Supplier Webpage Request for quotationCAS: 81334-34-1
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Imazapyr acid Request for quotation
CAS: 81334-34-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 81334-34-1
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Imazapyr acid Request for quotation
CAS: 81334-34-1
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 81334-34-1
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Multiple SpecificationsSpot supply
Product Name: Imazapyr Acid Visit Supplier Webpage Request for quotationCAS: 81334-34-1
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




