7803-65-8
Ammonium Hypophosphite
CAS: 7803-65-8
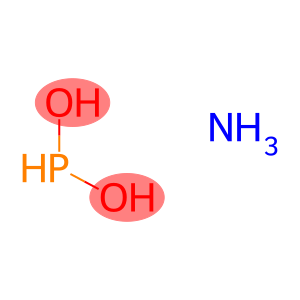
Molecular Formula: H3N.H3O2P
7803-65-8 - Names and Identifiers
| Name | Ammonium Hypophosphite |
| Synonyms | AMMONIUM PHOSPHINATE Ammonium phosphenite aMMoniuM phosphonite Ammonium hypophosphi AMMONIUM HYPOPHOSPHITE Ammonium Hypophosphite Phosphinic acid, ammonium salt hypophosphorousacid,ammoniumsalt AMMONIUM HYPOPHOSPHITE EXTRA PURE Ammonium hypophosphite, for analysis |
| CAS | 7803-65-8 |
| EINECS | 232-266-0 |
| InChI | InChI=1/H3N.HO2P/c;1-3-2/h1H3;(H,1,2) |
| InChIKey | ZRDXIILBYDTYOA-UHFFFAOYSA-N |
7803-65-8 - Physico-chemical Properties
| Molecular Formula | H3N.H3O2P |
| Molar Mass | 83.03 |
| Density | 1.634 [CRC10] |
| Melting Point | 155-160 °C |
| Water Solubility | 1000 g/L |
| Appearance | white hygroscopic crystals |
| Merck | 13,530 |
| Physical and Chemical Properties | Character: White rhombic flaky or granular crystal. melting point 200 ℃ relative density 1.634g/cm3 solubility soluble in water, alcohol, ammonia, insoluble in acetone. Its aqueous solution is neutral. |
| Use | Used in the manufacture of soft flux (welding stainless steel, etc.) and polyamide catalyst |
7803-65-8 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| HS Code | 28351000 |
7803-65-8 - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Overview | ammonium hypophosphite, also known as ammonium hypophosphite or ammonium hypophosphite, chemical formula NH4H2PO2, white flaky crystal, prismatic orthorhombic crystal system. Soluble in water, aqueous solution was neutral reaction, soluble in ethanol, ammonia, insoluble in acetone. Deliquescence. Upon heating to 240 °c, decomposition and evolution of phosphine occurred. Used as analytical reagents and in the pharmaceutical industry. |
| properties | ammonium hypophosphite is white flaky crystal with prismatic orthorhombic system; Melting Point: 200 ℃; Relative density (specific gravity):1.634; Soluble in water, soluble in ethanol, ammonia, difficult to dissolve in acetone, which dissolve 1g ammonium phosphate needs about 1ml water, 0.2ml boiling water, 20ml ethanol, the aqueous solution is neutral; Deliquescence; combustible phosphine is decomposed and evolved upon heating to 240 °c. |
| preparation method | 1. One-step method: the direct reaction of yellow phosphorus and ammonia, but the reaction conditions are difficult to control, often have a certain by-product ammonium phosphite generation, in addition, the method will also produce PH3 waste gas, PH3 gas toxic, burning in air is very dangerous. 2. Neutralization method: ammonia gas can be passed into high purity hypophosphorous acid to generate ammonium hypophosphite solution, and then through evaporation and crystallization to obtain ammonium hypophosphite finished product. The operation link of preparing hypophosphorous acid in this method is more complicated, so the operation of this method is more complicated. 3. Ammonium hypophosphite solution is prepared by adding ammonium carbonate, an ammonium salt containing acid groups capable of being precipitated by calcium ions, to calcium hypophosphite solution, and then concentrated to obtain ammonium hypophosphite solid. |
| Use | 1. Used as analytical reagent; 2. Used in pharmaceutical industry; 3. Used in aluminum electrolytic capacitor, polyamide. used to make soft flux (welded stainless steel, etc.) and polyamide catalyst used as analytical reagent, also used in pharmaceutical industry |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Ammonium Hypophosphite Request for quotationCAS: 7803-65-8
Tel: 86+027-83389957
Email: 1024042217@qq.com
Mobile: 86+18627766980
QQ: 1024042217
Wechat: 18627766980
Spot supply
Product Name: Ammonium hypophosphite Visit Supplier Webpage Request for quotationCAS: 7803-65-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: Ammonium hypophosphite Request for quotationCAS: 7803-65-8
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Multiple Specifications
Product Name: Ammonium hypophosphite Request for quotationCAS: 7803-65-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: Ammonium Hypophosphite Request for quotationCAS: 7803-65-8
Tel: 86+027-83389957
Email: 1024042217@qq.com
Mobile: 86+18627766980
QQ: 1024042217
Wechat: 18627766980
Spot supply
Product Name: Ammonium hypophosphite Visit Supplier Webpage Request for quotationCAS: 7803-65-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: Ammonium hypophosphite Request for quotationCAS: 7803-65-8
Tel: +86 19849939632
Email: 2470479589@qq.com
Mobile: +86 19849939632
QQ: 2470479589
Wechat: +86 19849939632
Multiple Specifications
Product Name: Ammonium hypophosphite Request for quotationCAS: 7803-65-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
View History



