7697-23-6
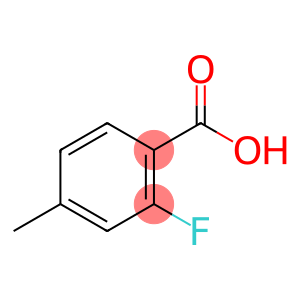
2-Fluoro-4-methylbenzoic acid
CAS: 7697-23-6
Molecular Formula: C8H7FO2
7697-23-6 - Names and Identifiers
| Name | 2-Fluoro-4-methylbenzoic acid |
| Synonyms | RARECHEM AL BO 2237 2-Fluoro-p-toluic Acid 4-Carboxy-3-fluorotoluene 2-FLUORO-4-METHYLBENZOIC ACID 2-Fluoro-4-methylbenzoic acid Benzoic acid, 2-fluoro-4-methyl- 2-Fluoro-4-methylbenzoic acid fandachem Benzene, 1,2,3,4,5-pentafluoro-6-methyl- 4-Carboxy-3-fluorotoluene, 2-Fluoro-p-toluic acid |
| CAS | 7697-23-6 |
| EINECS | 211-485-5 |
| InChI | InChI=1/C7H3F5/c1-2-3(8)5(10)7(12)6(11)4(2)9/h1H3 |
7697-23-6 - Physico-chemical Properties
| Molecular Formula | C8H7FO2 |
| Molar Mass | 154.14 |
| Density | 1.258±0.06 g/cm3(Predicted) |
| Melting Point | 186-189°C |
| Boling Point | 271.7±20.0 °C(Predicted) |
| Flash Point | 34.4°C |
| Solubility | soluble in Methanol |
| Vapor Presure | 21mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Light yellow to Light orange |
| pKa | 3.44±0.10(Predicted) |
| Storage Condition | Sealed in dry,Room Temperature |
| Refractive Index | 1.404 |
| MDL | MFCD03092906 |
7697-23-6 - Risk and Safety
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R22 - Harmful if swallowed |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S37 - Wear suitable gloves. |
| HS Code | 29163990 |
| Hazard Class | IRRITANT |
7697-23-6 - Reference Information
| Introduction | 2-fluoro-4-methylbenzoic acid is an extremely light yellow or light red-yellow solid powder at normal temperature and pressure, it is a benzoic acid derivative, which has the general properties of carboxylic acid. It is mainly used for the preparation of preservatives, and for the synthesis of drugs and dyes. |
| Use | 2-fluoro-4-methylbenzoic acid is mainly used for the preparation of preservatives, and dyes, it can also be used as pharmaceutical intermediates and organic synthesis intermediates. In the synthetic conversion, the carboxyl group in 2-fluoro-4-methylbenzoic acid can be esterified under acidic or basic conditions, and the carboxyl group can be converted into an amide group under the action of dichloro sulfoxide. |
| synthesis method | triazene derivatives (85.0 μmol) add to perfluorohexane (1 mL/100 mg) and slowly add the tetrafluoroborate ether complex (0.744 mmol) to the mixture with stirring, the vial was capped with a pressure-resistant jaw and the reaction mixture was stirred at 80°C for 12 hours. After cooling to room temperature, acetone was slowly added to the mixture, the solvent was evaporated (up to 40°C and 200 mbar), the mixture was shaken again and the acetone layer was separated. The fluorinated solvent was extracted again with acetone and the combined acetone layers were concentrated in vacuo by column chromatography (cyclohexane: ethyl acetate 10 to 1 to 1). The target product, 2-fluoro-4-methylbenzoic acid, was obtained by purification of the product. Fig.2-synthesis of fluoro-4-methylbenzoic acid |
Last Update:2024-04-09 19:05:15
Supplier List
Spot supply
Product Name: 2-Fluoro-4-methylbenzoic acid Visit Supplier Webpage Request for quotationCAS: 7697-23-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: 2-Fluoro-4-methylbenzoic acid Request for quotationCAS: 7697-23-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-Fluoro-4-Methylbenzoic Acid Visit Supplier Webpage Request for quotation
CAS: 7697-23-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7697-23-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Fluoro-4-methylbenzoic acid Visit Supplier Webpage Request for quotationCAS: 7697-23-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: 2-Fluoro-4-methylbenzoic acid Request for quotationCAS: 7697-23-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: 2-Fluoro-4-Methylbenzoic Acid Visit Supplier Webpage Request for quotation
CAS: 7697-23-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7697-23-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


