7481-89-2
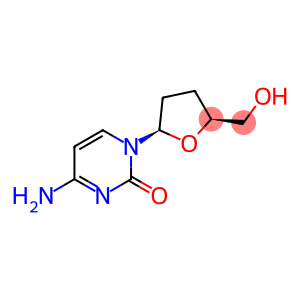
2',3'-dideoxycytidine
CAS: 7481-89-2
Molecular Formula: C9H13N3O3
7481-89-2 - Names and Identifiers
| Name | 2',3'-dideoxycytidine |
| Synonyms | DDC D 2C ZALCITABINE zalcitabine dideoxycytidine 2',3'-DIDEOXYCYTIDINE 2',3'-dideoxycytidine 2',3'-dideoxy-cytidin 2',3'-Dideoxy-D-cytidine BETA-D-2',3'-DIDEOXYCYTIDINE Cytidine, 2',3'-dideoxy- (8CI, 9CI) 2'',3''-DIDEOXYCYTIDINE(ZALCITABINE) 1-(2',3'-DIDEOXY-BETA-RIBOFURANOSYL)CYTOSINE 4-AMINO-1-[(2R,5S)-5-(HYDROXYMETHYL)OXOLAN-2-YL]PYRIMIDIN-2-ONE 4-amino-1-[5-(hydroxymethyl)tetrahydrofuran-2-yl]pyrimidin-2(1H)-one 4-amino-1-[(5S)-5-(hydroxymethyl)tetrahydrofuran-2-yl]pyrimidin-2(1H)-one |
| CAS | 7481-89-2 |
| EINECS | 620-762-3 |
| InChI | InChI=1/C9H13N3O3/c10-7-3-4-12(9(14)11-7)8-2-1-6(5-13)15-8/h3-4,6,8,13H,1-2,5H2,(H2,10,11,14)/t6-,8?/m0/s1 |
| InChIKey | WREGKURFCTUGRC-POYBYMJQSA-N |
7481-89-2 - Physico-chemical Properties
| Molecular Formula | C9H13N3O3 |
| Molar Mass | 211.22 |
| Density | 1.2605 (rough estimate) |
| Melting Point | 217-218°C(lit.) |
| Boling Point | 350.9°C (rough estimate) |
| Specific Rotation(α) | D25 +81° (c = 0.635 in water) |
| Flash Point | 204.776°C |
| Water Solubility | 5-10 g/100 mL at 19 ºC |
| Solubility | DMSO (Slightly, Heated), Methanol (Slightly), Water (Slightly, Sonicated) |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | powder |
| Color | colorless |
| Merck | 14,10109 |
| BRN | 654956 |
| pKa | 14.44±0.10(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents. |
| Refractive Index | 78 ° (C=0.5, H2O) |
| MDL | MFCD00012188 |
| Physical and Chemical Properties | Crystallization from ethanol and benzene, melting point 215-217 °c melting point 209-210 °c has also been reported. [Α] D23 8l °(C = 0.635, water). UV maximum absorption (0.1mol/L hydrochloric acid):280nm (× 17720);(0.1mol/L sodium hydroxide):270nm (× 8410). |
7481-89-2 - Risk and Safety
| Risk Codes | R40 - Limited evidence of a carcinogenic effect R36/37 - Irritating to eyes and respiratory system. R34 - Causes burns |
| Safety Description | S22 - Do not breathe dust. S36 - Wear suitable protective clothing. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S27 - Take off immediately all contaminated clothing. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | HA3870000 |
| FLUKA BRAND F CODES | 10-23 |
| HS Code | 2934990002 |
7481-89-2 - Reference
| Reference Show more | 1. [IF=4.411] Yujiao Hua et al."Quality Evaluation of Pseudostellariae Radix Based on Simultaneous Determination of Multiple Bioactive Components Combined with Grey Relational Analysis."Molecules. 2017 Jan;22(1):13 2. [IF=1.618] Wang Shengnan et al."Simultaneous Determination of Iridoid Glycosides, Phenylpropanoid Glycosides, Organic Acids, Nucleosides and Amino Acids in Scrophulariae Radix Processed by Different Processing Methods by HPLC-QTRAP-MS/MS."J Chromatogr Sci. 2021 Jun; |
7481-89-2 - Reference Information
| (IARC) carcinogen classification | 2B (Vol. 76) 2000 |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| biological activity | zallitabine (NSC 606170, Ro 24-2027/000) is a nucleoside HIV reverse transcriptase inhibitor (NARTI). |
| Target | Value |
| Use | acts at the early stage of HIV replication, inhibiting Virus of reverse transcriptase and terminating Virus of DNA strand replication. For the treatment of patients with advanced immunodeficiency virus infection. antiviral drugs, used for anti-cancer, anti-HIV |
| production method | N-acetyl cytosine nucleoside (4G, 14 mmol) was refluxed in mL of acetonitrile, acetyl bromide (10ml,0.137 mol) was added dropwise over 30min and refluxed for 3h after addition. The solvent was distilled off, and the residue was dissolved in 100ml of chloroform and washed with 100ml of water. The chloroform was distilled off, and the residue was recrystallized from 10ml of ethanol to obtain 2g of compound (I) in a yield of 33% with a melting point of 79 ° -180 ° C. (decomposition) as colorless Flake crystals. Compound (I)(1G, 2.3 mmol), 1g of calcium carbonate, 50% G of palladium-barium sulfate and 50ml of methanol were shaken with hydrogen gas for 1H at 25 ° C. And normal pressure (60ml of hydrogen was absorbed). The crystals were removed by filtration and the filtrate was concentrated to dryness. The residue was dissolved in 10ml of a 20% ammonia-methanol solution and allowed to stand at 5 °c overnight. After column chromatography, it was recrystallized from ethanol to obtain 34mg of colourless crystalline gemcitabine in a yield of 7%. |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 2′,3′-Dideoxycytidine Visit Supplier Webpage Request for quotationCAS: 7481-89-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: Zalcitabine Request for quotationCAS: 7481-89-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Zalcitabine Request for quotation
CAS: 7481-89-2
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 7481-89-2
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Spot supply
Product Name: 2′,3′-Dideoxycytidine Visit Supplier Webpage Request for quotationCAS: 7481-89-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple Specifications
Product Name: 2′,3′-Dideoxycytidine Request for quotationCAS: 7481-89-2
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog
Spot supply
Product Name: 2′,3′-Dideoxycytidine Visit Supplier Webpage Request for quotationCAS: 7481-89-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: Zalcitabine Request for quotationCAS: 7481-89-2
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Zalcitabine Request for quotation
CAS: 7481-89-2
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 7481-89-2
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Spot supply
Product Name: 2′,3′-Dideoxycytidine Visit Supplier Webpage Request for quotationCAS: 7481-89-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple Specifications
Product Name: 2′,3′-Dideoxycytidine Request for quotationCAS: 7481-89-2
Tel: +86-571-88162785
Email: Lucy@verypharm.com
Mobile: +86-13606544505
QQ: 472954302
Product List: View Catalog
View History




