73852-18-3
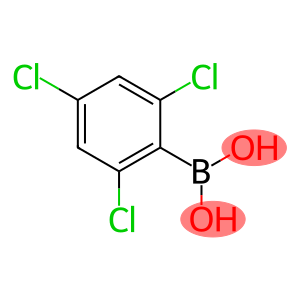
2,4,6-Trichlorophenylboronic acid
CAS: 73852-18-3
Molecular Formula: C6H4BCl3O2
73852-18-3 - Names and Identifiers
73852-18-3 - Physico-chemical Properties
| Molecular Formula | C6H4BCl3O2 |
| Molar Mass | 225.26 |
| Density | 1.60±0.1 g/cm3(Predicted) |
| Melting Point | 162-164 |
| Boling Point | 363.9±52.0 °C(Predicted) |
| Flash Point | 173.9°C |
| Solubility | soluble in Methanol |
| Vapor Presure | 6.22E-06mmHg at 25°C |
| Appearance | powder to crystal |
| Color | White to Almost white |
| pKa | 7.80±0.58(Predicted) |
| Storage Condition | Inert atmosphere,2-8°C |
| Refractive Index | 1.594 |
| MDL | MFCD01074621 |
73852-18-3 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Hazard Note | Harmful/Irritant/Keep Cold |
73852-18-3 - Introduction
2,4, 6-Trichorylboronic acid is an organic compound with the chemical formula C6H3Cl3BO2. It is a white crystalline solid, hygroscopic at room temperature. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: White crystalline solid
-Molecular weight: 251.33g/mol
-melting point: 179-182 ℃
-Solubility: High solubility in non-polar solvents (such as ether), difficult to dissolve in water
-Stability: relatively stable at room temperature, but may decompose under high temperature, high humidity or light conditions
Use:
2,4,6-Trichlorophenylboronic acid is mainly used in organic synthesis reactions, especially as an important derivative of ligands in metal ligand catalyzed reactions. It can be used as a precursor of tertiary amine ligands, and palladium and other metal coordination to form a stable complex, used in C- C bond formation reaction, reduction reaction and C- H bond functionalization reaction. In addition, it can also be used to prepare pharmaceutical intermediates, photoelectric materials, etc.
Preparation Method:
The preparation method of 2,4,6-Trichlorophenylboronic acid is generally obtained by reacting 2,4, 6-trichlorophenylboronic compound with acid. Common synthetic methods include the reaction of boric acid with 2,4, 6-trichlorophenylmagnesium solution, or the hydrolysis of 2,4, 6-Tricholophenylboronic acid ester to obtain the target product.
Safety Information:
- 2,4, 6-Trichorophenylboronic acid is an organic compound and should comply with general laboratory safety practices.
-Wear appropriate personal protective equipment, such as lab gloves, glasses, and lab coats.
-Avoid direct contact with skin, eyes and respiratory tract.
-Use should be operated in a well-ventilated place to avoid inhaling its steam or dust.
-in storage, should be sealed in a cool, dry place, away from heat and fire.
-Detailed toxicological data and safety information can be obtained from chemical suppliers or relevant databases. When using or handling this compound, please follow the relevant safety guidelines and regulations.
Nature:
-Appearance: White crystalline solid
-Molecular weight: 251.33g/mol
-melting point: 179-182 ℃
-Solubility: High solubility in non-polar solvents (such as ether), difficult to dissolve in water
-Stability: relatively stable at room temperature, but may decompose under high temperature, high humidity or light conditions
Use:
2,4,6-Trichlorophenylboronic acid is mainly used in organic synthesis reactions, especially as an important derivative of ligands in metal ligand catalyzed reactions. It can be used as a precursor of tertiary amine ligands, and palladium and other metal coordination to form a stable complex, used in C- C bond formation reaction, reduction reaction and C- H bond functionalization reaction. In addition, it can also be used to prepare pharmaceutical intermediates, photoelectric materials, etc.
Preparation Method:
The preparation method of 2,4,6-Trichlorophenylboronic acid is generally obtained by reacting 2,4, 6-trichlorophenylboronic compound with acid. Common synthetic methods include the reaction of boric acid with 2,4, 6-trichlorophenylmagnesium solution, or the hydrolysis of 2,4, 6-Tricholophenylboronic acid ester to obtain the target product.
Safety Information:
- 2,4, 6-Trichorophenylboronic acid is an organic compound and should comply with general laboratory safety practices.
-Wear appropriate personal protective equipment, such as lab gloves, glasses, and lab coats.
-Avoid direct contact with skin, eyes and respiratory tract.
-Use should be operated in a well-ventilated place to avoid inhaling its steam or dust.
-in storage, should be sealed in a cool, dry place, away from heat and fire.
-Detailed toxicological data and safety information can be obtained from chemical suppliers or relevant databases. When using or handling this compound, please follow the relevant safety guidelines and regulations.
Last Update:2024-04-09 02:00:47
Supplier List
Spot supply
Product Name: 2,4,6-Trichlorophenylboronic acid Visit Supplier Webpage Request for quotationCAS: 73852-18-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: 2,4,6-Trichlorophenylboronic acid Request for quotationCAS: 73852-18-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 2,4,6-Trichlorobenzeneboronic acid Visit Supplier Webpage Request for quotationCAS: 73852-18-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2,4,6-Trichlorophenylboronic acid Visit Supplier Webpage Request for quotationCAS: 73852-18-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: 2,4,6-Trichlorophenylboronic acid Request for quotationCAS: 73852-18-3
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 2,4,6-Trichlorobenzeneboronic acid Visit Supplier Webpage Request for quotationCAS: 73852-18-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


