7340-22-9
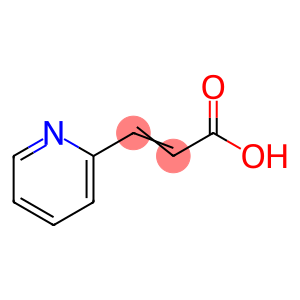
3-(2-Pyridyl)acrylic acid
CAS: 7340-22-9
Molecular Formula: C8H7NO2
7340-22-9 - Names and Identifiers
| Name | 3-(2-Pyridyl)acrylic acid |
| Synonyms | RARECHEM AH CK 0188 2-Pyridineacrylic acid 3-(2-Pyridyl)acrylic acid 3-(PYRID-2-YL)ACRYLIC ACID 3-PYRIDIN-2-YL-ACRYLIC ACID 3-(Pyridin-2-yl)acrylic acid (E)-3-(Pyridin-2-yl)acrylic acid (2E)-3-(Pyridin-2-yl)acrylic acid 3-(pyridin-2-yl)prop-2-enoic acid (2E)-3-(pyridin-2-yl)prop-2-enoic acid |
| CAS | 7340-22-9 |
| EINECS | 214-424-0 |
| InChI | InChI=1/C8H7NO2/c10-8(11)5-4-7-3-1-2-6-9-7/h1-6H,(H,10,11)/b5-4- |
7340-22-9 - Physico-chemical Properties
| Molecular Formula | C8H7NO2 |
| Molar Mass | 149.15 |
| Density | 1.261±0.06 g/cm3(Predicted) |
| Melting Point | 201-203°C |
| Boling Point | 304.8±17.0 °C(Predicted) |
| Flash Point | 138.1°C |
| Vapor Presure | 0.000376mmHg at 25°C |
| pKa | 2.06±0.10(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Refractive Index | 1.624 |
7340-22-9 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | 36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. |
| Hazard Note | Irritant |
7340-22-9 - Introduction
3-(2-Pyridyl)acrylic acid, chemical formula C8H7NO2, is an organic compound. The following is a description of its nature, use, preparation and safety information:
Nature:
3-(2-Pyridyl)acrylic acid is a white or yellowish solid, stable at room temperature. It is a harmless compound that dissolves in water.
Use:
3-(2-Pyridyl)acrylic acid is widely used in organic synthesis and pharmaceutical chemistry. It can be used as a synthetic intermediate for the synthesis of different types of organic compounds, such as drugs containing pyridine structure, complexes and so on.
Preparation Method:
The preparation of 3-(2-Pyridyl)acrylic acid generally adopts the method of chemical synthesis. A common preparation method is through the carboxylation reaction of pyridine, followed by esterification reaction to obtain 3-(2-Pyridyl)acrylic acid.
Safety Information:
3-(2-Pyridyl)acrylic acid is generally safe under normal conditions of use. However, as a chemical, it can be irritating to the skin, eyes and respiratory system. Therefore, when handling 3-(2-Pyridyl)acrylic acid, it is recommended to take appropriate personal protective measures, such as wearing protective gloves, goggles and masks. At the same time, it should also be kept away from open flames and oxidants, and stored in a dry, well-ventilated place. It should not be mixed with strong acids, strong bases and oxidants.
Nature:
3-(2-Pyridyl)acrylic acid is a white or yellowish solid, stable at room temperature. It is a harmless compound that dissolves in water.
Use:
3-(2-Pyridyl)acrylic acid is widely used in organic synthesis and pharmaceutical chemistry. It can be used as a synthetic intermediate for the synthesis of different types of organic compounds, such as drugs containing pyridine structure, complexes and so on.
Preparation Method:
The preparation of 3-(2-Pyridyl)acrylic acid generally adopts the method of chemical synthesis. A common preparation method is through the carboxylation reaction of pyridine, followed by esterification reaction to obtain 3-(2-Pyridyl)acrylic acid.
Safety Information:
3-(2-Pyridyl)acrylic acid is generally safe under normal conditions of use. However, as a chemical, it can be irritating to the skin, eyes and respiratory system. Therefore, when handling 3-(2-Pyridyl)acrylic acid, it is recommended to take appropriate personal protective measures, such as wearing protective gloves, goggles and masks. At the same time, it should also be kept away from open flames and oxidants, and stored in a dry, well-ventilated place. It should not be mixed with strong acids, strong bases and oxidants.
Last Update:2024-04-09 21:11:58
Supplier List
Spot supply
Product Name: 3-(Pyridin-2-yl)acrylic acid Visit Supplier Webpage Request for quotationCAS: 7340-22-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: 3-(2-PYRIDYL)ACRYLIC ACID Request for quotationCAS: 7340-22-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 3-(2-PYRIDYL)ACRYLIC ACID Visit Supplier Webpage Request for quotationCAS: 7340-22-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 3-(Pyridin-2-yl)acrylic acid Visit Supplier Webpage Request for quotationCAS: 7340-22-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: 3-(2-PYRIDYL)ACRYLIC ACID Request for quotationCAS: 7340-22-9
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: 3-(2-PYRIDYL)ACRYLIC ACID Visit Supplier Webpage Request for quotationCAS: 7340-22-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


