717927-65-6
trichloroiridium hydrochloride hydrate
CAS: 717927-65-6
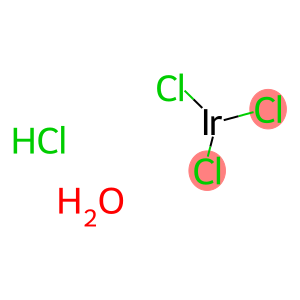
Molecular Formula: IrCl3xH2OyHCl
717927-65-6 - Names and Identifiers
| Name | trichloroiridium hydrochloride hydrate |
| Synonyms | trichloroiridium hydrochloride hydrate trichloroiridium,hydrate,hydrochloride Iridium(III) chloride hydrate hydrochloride |
| CAS | 717927-65-6 |
| EINECS | 684-838-8 |
| InChI | InChI=1/4ClH.Ir.H2O/h4*1H;;1H2/q;;;;+3;/p-3 |
717927-65-6 - Physico-chemical Properties
| Molecular Formula | IrCl3xH2OyHCl |
| Molar Mass | 353.05 |
| Density | 5.3g/mLat 25°C(lit.) |
| Appearance | powder |
| Storage Condition | Room Temprature |
717927-65-6 - Risk and Safety
| Risk Codes | R34 - Causes burns R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36 - Wear suitable protective clothing. |
| UN IDs | UN 3261 8/PG 2 |
| WGK Germany | 3 |
717927-65-6 - Introduction
trichloroiridium hydrochloride hydrate is a chemical compound with the formula IrCl3 · HCl · xH2O. Here are some of its properties, uses, methods and safety information:
Nature:
1. Appearance: trichloroiridium hydrochloride hydrate are usually colorless or white crystals.
2. Stability: It has good thermal stability and is relatively stable at room temperature.
3. Solubility: It can be dissolved in water to form an acidic solution.
Use:
1. Catalyst: trichloroiridium hydrochloride hydrate can be used as catalysts for many chemical reactions, such as oxidation reactions, addition reactions, etc.
2. Electrochemical applications: It is also commonly used in the preparation of electrochemical sensors and fuel cells and other electrochemical devices.
Preparation Method:
The preparation method of trichloroiridium hydrochloride hydrate can be carried out by the following steps:
1. First, iridium metal is reacted with excess hydrochloric acid to generate iridium trichloride.
2. Next, iridium trichloride is reacted with hydrochloric acid to obtain iridium trichloride hydrochloride.
3. Finally, iridium trichloride hydrochloride was crystallized using water as a solvent to obtain a hydrate.
Safety Information:
1. This compound is irritating to skin, eyes and mucous membranes and should be avoided when in contact.
2. It should be stored in a dry, well-ventilated place, and away from combustibles and oxidants.
3. Wear appropriate laboratory protective equipment such as gloves, goggles, and laboratory coats when handling trichloroiridium hydrochloride hydrate. In case of any accident, corresponding first aid measures shall be taken in time.
Nature:
1. Appearance: trichloroiridium hydrochloride hydrate are usually colorless or white crystals.
2. Stability: It has good thermal stability and is relatively stable at room temperature.
3. Solubility: It can be dissolved in water to form an acidic solution.
Use:
1. Catalyst: trichloroiridium hydrochloride hydrate can be used as catalysts for many chemical reactions, such as oxidation reactions, addition reactions, etc.
2. Electrochemical applications: It is also commonly used in the preparation of electrochemical sensors and fuel cells and other electrochemical devices.
Preparation Method:
The preparation method of trichloroiridium hydrochloride hydrate can be carried out by the following steps:
1. First, iridium metal is reacted with excess hydrochloric acid to generate iridium trichloride.
2. Next, iridium trichloride is reacted with hydrochloric acid to obtain iridium trichloride hydrochloride.
3. Finally, iridium trichloride hydrochloride was crystallized using water as a solvent to obtain a hydrate.
Safety Information:
1. This compound is irritating to skin, eyes and mucous membranes and should be avoided when in contact.
2. It should be stored in a dry, well-ventilated place, and away from combustibles and oxidants.
3. Wear appropriate laboratory protective equipment such as gloves, goggles, and laboratory coats when handling trichloroiridium hydrochloride hydrate. In case of any accident, corresponding first aid measures shall be taken in time.
Last Update:2024-04-10 22:29:15
Supplier List
Product Name: IRIDIUM(III) CHLORIDE HYDROCHLORIDE HYD& Request for quotation
CAS: 717927-65-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 717927-65-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Iridium(Iii) Chloride HydROChloride Hyd& Visit Supplier Webpage Request for quotation
CAS: 717927-65-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 717927-65-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: IRIDIUM(III) CHLORIDE HYDROCHLORIDE HYD& Request for quotation
CAS: 717927-65-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 717927-65-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Iridium(Iii) Chloride HydROChloride Hyd& Visit Supplier Webpage Request for quotation
CAS: 717927-65-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 717927-65-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

