71626-98-7
Calcium iodide hydrate
CAS: 71626-98-7
Molecular Formula: CaH2I2O
71626-98-7 - Names and Identifiers
71626-98-7 - Physico-chemical Properties
| Molecular Formula | CaH2I2O |
| Molar Mass | 311.9 |
| Density | 2.55 |
| Melting Point | 783°C |
| Boling Point | 160°C |
| Water Solubility | Freely soluble in water and alcohol |
| Appearance | Lumps |
| Color | Yellow |
| Merck | 14,1677 |
| Sensitive | Hygroscopic |
| Physical and Chemical Properties | Colorless or yellow-white crystal or powder, bitter taste, deliquescent. Relative density 2.55. Melting point 42 ℃ (decomposition). Boiling point 160 ℃. Easily soluble in water, slightly soluble in ethanol and acetone. Its aqueous solution is neutral, soluble in acid and decomposed to free iodine or generate hydroiodic acid. Together with ammonium iodide, it is heated and dehydrated in a stream of hydrogen iodide or dry nitrogen to produce anhydrous calcium iodide. |
71626-98-7 - Risk and Safety
| Risk Codes | 61 - May cause harm to the unborn child |
| Safety Description | S22 - Do not breathe dust. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| WGK Germany | 3 |
| TSCA | Yes |
| Toxicity | 参见碘。 |
71626-98-7 - Reference Information
| toxicity | see iodine. |
| chemical properties | colorless or yellow-white crystal or powder, bitter taste, deliquescent. Relative density 2.55. Melting point 42 ℃ (decomposition). Boiling point 160 ℃. Easily soluble in water, slightly soluble in ethanol and acetone. Its aqueous solution is neutral, soluble in acid and decomposed to free iodine or generate hydroiodic acid. Together with ammonium iodide, it is heated and dehydrated in a stream of hydrogen iodide or dry nitrogen to produce anhydrous calcium iodide. |
| use | is mainly used as medicine and is a substitute for potassium iodide. Used as a photosensitive emulsion for photographic film and as a desiccant for hydrogen iodide. The fire industry is used to prepare fire extinguishing agents. It is also used to make mineral water. Mainly used in medicine, as a substitute for potassium iodide; photography is used as a photosensitive emulsion; it can also be used as a desiccant and analytical reagent for making mineral water, fire extinguishing agent, hydrogen iodide. |
| production method | direct synthesis method according to the weight ratio of iodine: sulfuric acid: sodium sulfide: calcium oxide: distilled water = 1:0.75:0.75:1:3.3, sodium sulfide is first added with water to make 30% solution, and then 50% dilute sulfuric acid is dropped under stirring, to slightly acidic (pH = 6), the generated hydrogen sulfide gas is washed with distilled water. Iodine is added to distilled water, hydrogen sulfide is introduced while stirring, the absorber is cooled with water, the pressure is kept below 0.049MPa, the absorption temperature is 25~30 ℃, the iodine is completely dissolved, and it reacts with hydrogen sulfide to generate milky white hydrogen iodide solution. After filtering, the prefabricated calcium hydroxide is added until alkaline (pH7.5 ~ 8), and after filtering, the calcium iodide solution is obtained. Add activated carbon for decolorization, filtration, heating to 200 ℃ for concentration, then dilute with water to 1.54 relative density, and let stand overnight. Filter, then heat to 218~230 ℃ for evaporation and concentration, quickly cool with ice water (the speed should be fast, otherwise it will turn yellow), after crushing, the finished product of calcium iodide is prepared. The H2S + I2 → 2HI + S2HI + Ca(OH)2 → CaI2 + 2H2O residue is washed twice with an appropriate amount of distilled water, and the washing liquid and filtrate are combined and refined. The ferrous iodide method adds distilled water to the reactor, adds the iron filings that have been removed from the oil under stirring, and adds iodine in stages. During the reaction, the temperature is controlled not to exceed 40°C until the iodine is completely dissolved, and then add distilled water. Heat to boiling, so that iodine and iron filings fully react. Then add slaked lime to a pH of 14. Heat to boiling and stir for 1h to fully react ferrous iodide and calcium hydroxide to produce calcium iodide. Cool to 50 ℃, release the material, and obtain the filtrate after centrifugal separation, then mix the filter cake with twice the amount of water, boil, cool and centrifuge, and combine the two filtrates for refining. The filtrate was adjusted to pH 2~3 with hydroiodic acid, evaporated, concentrated, cooled and crushed at 220~230 ℃ to prepare calcium iodide finished product. Its I2 + Fe → FeI2Fel2 + Ca(OH)2 → CaI2 + Fe(OH)2 |
Last Update:2024-04-10 22:29:15
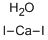
Supplier List
Spot supply
Product Name: Calcium iodide hydrate Visit Supplier Webpage Request for quotationCAS: 71626-98-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: CALCIUM IODIDE HYDRATE Request for quotation
CAS: 71626-98-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 71626-98-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Spot supply
Product Name: Calcium iodide hydrate Visit Supplier Webpage Request for quotationCAS: 71626-98-7
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: CALCIUM IODIDE HYDRATE Request for quotation
CAS: 71626-98-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 71626-98-7
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
View History

