714971-28-5
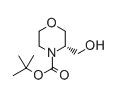
tert-Butyl (3S)-3-(hydroxymethyl)morpholine-4-carboxylate
CAS: 714971-28-5
Molecular Formula: C10H19NO4
714971-28-5 - Names and Identifiers
714971-28-5 - Physico-chemical Properties
| Molecular Formula | C10H19NO4 |
| Molar Mass | 217.26 |
| Density | 1.118 |
| Melting Point | 80 ºC |
| Boling Point | 320.7±27.0 °C(Predicted) |
| Flash Point | 147.8°C |
| Water Solubility | Slightly soluble in water. |
| Vapor Presure | 2.53E-05mmHg at 25°C |
| Appearance | Powder |
| Color | White |
| pKa | 14.85±0.10(Predicted) |
| Storage Condition | Sealed in dry,Store in freezer, under -20°C |
| Refractive Index | 1.474 |
714971-28-5 - Reference Information
| use | (3S)-3-(hydroxymethyl) morpholin-4-carboxylate tert-butyl ester is a key intermediate in the synthesis of BMS-599626, BMS-599626 selective and effective oral inhibitors of human epidermal growth factor receptor 1 and 2 kinases. As an organic chemical intermediate, N-substituted morpholine has the dual properties of tertiary amine and ether, and has a wide range of applications. It can be used as a catalyst for the production of polyurethane foam. The catalyst in the synthesis process can also be used as a surfactant, extractant, corrosion inhibitor, and its derivative N-methyl morpholine oxide. It is a very ideal spinning solvent for the manufacture of Lyocell natural fibers and Newcell man-made fiber filaments. |
| preparation | N-substituted morpholine contains double groups of tertiary amine and ether, which is not only an important catalyst in the foaming process of semi-synthetic penicillin and polyurethane, but also an important raw material for the synthesis of N-substituted morpholine oxide. The current synthesis process mainly includes morpholine method, N-methyl diethanolamine method, diethanolamine method, diethylene glycol method and dichloroether method. (3S)-3-(hydroxymethyl) morpholine -4-carboxylic acid tert-butyl ester is substituted with (R)-(4-benzyl-3-morpholine)-methanol as the starting material and is prepared by reacting with diptert-butyl dicarbonate. The synthesis reaction formula is as follows: Figure 1 (3S)-3-(hydroxymethyl) morpholine -4-carboxylic acid tert-butyl ester synthesis reaction experimental operation: add triethylamine to the dichloromethane solution of (R)-(4-benzyl-3-morpholine)-methanol, drop the dichloromethane solution of di-tert-butyl dicarbonate, drop it, and react for 15 hours. The solvent is distilled under reduced pressure, and the concentrate is processed by silica gel column chromatography [eluent: V (ethyl acetate) /V (petroleum ether) = 1/10] purified compound (3S)-3-(hydroxymethyl) morpholin-4-carboxylic acid tert-butyl ester. |
Last Update:2024-04-09 15:16:35
Supplier List
Spot supply
Product Name: (S)-4-Boc-(3-hydroxymethyl)morpholine Visit Supplier Webpage Request for quotationCAS: 714971-28-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: 3(S)-HYDROXYMETHYL-4-BOCMORPHOLINE Request for quotationCAS: 714971-28-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: (S)-3-Hydroxymethylmorpholine-4-carboxylic Acid tert-Butyl Ester Visit Supplier Webpage Request for quotation
CAS: 714971-28-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 714971-28-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (S)-4-Boc-(3-hydroxymethyl)morpholine Visit Supplier Webpage Request for quotationCAS: 714971-28-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: 3(S)-HYDROXYMETHYL-4-BOCMORPHOLINE Request for quotationCAS: 714971-28-5
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: (S)-3-Hydroxymethylmorpholine-4-carboxylic Acid tert-Butyl Ester Visit Supplier Webpage Request for quotation
CAS: 714971-28-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 714971-28-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


