709031-29-8
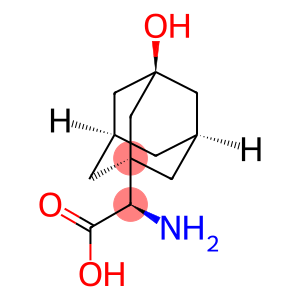
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7~]dec-1-yl)ethanoic acid
CAS: 709031-29-8
Molecular Formula: C12H19NO3
709031-29-8 - Names and Identifiers
| Name | (2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7~]dec-1-yl)ethanoic acid |
| Synonyms | Saxagliptin INT 1 Saxagliptin Intermediate 4 Saxagliptin Intermediate 1 (S)-3-hydroxyadamantylglycine 3-Hydroxy-1-adaMantyl-D-glyci 3-Hydroxy-1-adamantyl-D-glycine (2S)-2-AMino-2-(3-hydroxyadaMantan-1-yl)acetic acid (2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7~]dec-1-yl)ethanoic acid (alphaS)-alpha-Amino-3-hydroxytricyclo[3.3.1.13,7]decane-1-acetic acid Tricyclo[3.3.1.13,7]decane-1-acetic acid,alpha-amino-3-hydroxy-,(alphaS)- Tricyclo[3.3.1.13,7]decane-1-acetic acid,alpha-amino-3-hydroxy-, (alphaS)- |
| CAS | 709031-29-8 |
| EINECS | 1308068-626-2 |
| InChI | InChI=1/C12H19NO3/c13-9(10(14)15)11-2-7-1-8(3-11)5-12(16,4-7)6-11/h7-9,16H,1-6,13H2,(H,14,15)/t7?,8?,9-,11?,12?/m1/s1 |
709031-29-8 - Physico-chemical Properties
| Molecular Formula | C12H19NO3 |
| Molar Mass | 225.28 |
| Density | 1.409 |
| Melting Point | >260°C (dec.) |
| Boling Point | 402.0±20.0 °C(Predicted) |
| Flash Point | 196.903°C |
| Solubility | DMSO (Slightly, Heated), Water (Sparingly) |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | Solid |
| Color | Off-White |
| pKa | 2.36±0.10(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2–8 °C |
| Refractive Index | 1.645 |
709031-29-8 - Introduction
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid is an organic compound with the chemical formula C11H19NO3. The following is a description of the properties, uses, preparation and safety information of this compound:
Nature:
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid is a white solid with weak acidity. It is a chiral molecule that can exist in two conformations, D-and L-. It is soluble in water and some organic solvents and is a neutral solution.
Use:
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid is widely used in chemistry and biochemistry as chiral mass calibrators, enzyme catalysts and biological inhibitors. It can also be used in organic synthesis reactions, such as constructing chiral molecules, synthesizing drugs and regulating enzyme activities.
Preparation Method:
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid can be prepared by chemical synthesis or biotransformation. The chemical synthesis method includes using lithium adamantyl and acryloyl chloride to obtain 3-hydroxy-1-adamantylacetone, and then reacting with glycine to obtain the target product. Bioconversion methods can use microbial or enzymatic reactions to convert appropriate substrates into (2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid.
Safety Information:
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid is generally considered to be relatively safe. However, basic laboratory safety practices such as wearing gloves, safety glasses and laboratory protective clothing are still to be observed during handling and use. In addition, care should be taken to avoid contact with chemicals such as strong oxidants and strong acids, and to avoid inhaling their dust or solutions, so as not to cause irritation or allergic reactions. Please refer to the relevant safety data sheet for specific safety operation instructions.
Nature:
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid is a white solid with weak acidity. It is a chiral molecule that can exist in two conformations, D-and L-. It is soluble in water and some organic solvents and is a neutral solution.
Use:
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid is widely used in chemistry and biochemistry as chiral mass calibrators, enzyme catalysts and biological inhibitors. It can also be used in organic synthesis reactions, such as constructing chiral molecules, synthesizing drugs and regulating enzyme activities.
Preparation Method:
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid can be prepared by chemical synthesis or biotransformation. The chemical synthesis method includes using lithium adamantyl and acryloyl chloride to obtain 3-hydroxy-1-adamantylacetone, and then reacting with glycine to obtain the target product. Bioconversion methods can use microbial or enzymatic reactions to convert appropriate substrates into (2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid.
Safety Information:
(2S)-amino(3-hydroxytricyclo[3.3.1.1~3,7 ~]dec-1-yl)ethanoic acid is generally considered to be relatively safe. However, basic laboratory safety practices such as wearing gloves, safety glasses and laboratory protective clothing are still to be observed during handling and use. In addition, care should be taken to avoid contact with chemicals such as strong oxidants and strong acids, and to avoid inhaling their dust or solutions, so as not to cause irritation or allergic reactions. Please refer to the relevant safety data sheet for specific safety operation instructions.
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: Saxagliptin Impurity 29 Visit Supplier Webpage Request for quotationCAS: 709031-29-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: (2S)-2-Amino-2-(3-hydroxyadamantan-1-yl)acetic acid Visit Supplier Webpage Request for quotationCAS: 709031-29-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: (alphaS)-alpha-Amino-3-hydroxytricyclo[3.3.1.13,7]decane-1-acetic acid Request for quotationCAS: 709031-29-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: (alphaS)-alpha-Amino-3-hydroxytricyclo[3.3.1.13,7]decane-1-acetic acid Visit Supplier Webpage Request for quotation
CAS: 709031-29-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 709031-29-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Saxagliptin Impurity 29 Visit Supplier Webpage Request for quotationCAS: 709031-29-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Spot supply
Product Name: (2S)-2-Amino-2-(3-hydroxyadamantan-1-yl)acetic acid Visit Supplier Webpage Request for quotationCAS: 709031-29-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Multiple Specifications
Product Name: (alphaS)-alpha-Amino-3-hydroxytricyclo[3.3.1.13,7]decane-1-acetic acid Request for quotationCAS: 709031-29-8
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: (alphaS)-alpha-Amino-3-hydroxytricyclo[3.3.1.13,7]decane-1-acetic acid Visit Supplier Webpage Request for quotation
CAS: 709031-29-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 709031-29-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



