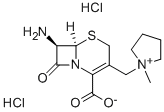
| overview | cefepime intermediate is the parent core of cefepime hydrochloride. At 0-5 ℃, the cefepime intermediate is dissolved in the solvent, and AE-active ester and triethylamine are added in a small amount to the whole reaction system. After the reaction, cefepime hydrochloride is obtained. |
| application | cefepime hydrochloride is the fourth generation cephalosporin for injection, developed by Bumai-Squibb (Bristol-MyersSquibb), and has been used for the treatment of various bacterial infectious diseases since it was first marketed in Sweden in 1993. Cefepime has a broad antibacterial spectrum. It not only maintains a good antibacterial effect against gram-negative bacteria, but also has strong anti-staphylococcal activity, strong antibacterial effect, high stability to β-lactamase, and good drug tolerance. Therefore, it is a promising antibiotic. |
| preparation | (1)7-ACA is dissolved in anhydrous cyclohexane, hexamethyldisilamine and trimethyliodosilane are added to vacuum reflux at 55 ℃ for 12h, cooling and dilution, dropping N-methylpyrrolidone, controlling the temperature to be less than 5 ℃, adding trimethyliodosilane, stirring for 30min, gradually raise the temperature to 40 ℃ and continue stirring for 23h;(2) Add anhydrous cyclohexane and methanol, keep the whole reaction system at 0-5 ℃ and stir for 15min, filter and wash to obtain crude product;(3) Add hydrochloric acid to the prepared crude product and stir, decolorize and filter with activated carbon, stir for 1h at 0-5 ℃, wash, and vacuum dry to obtain cefepime intermediate. |