66863-43-2
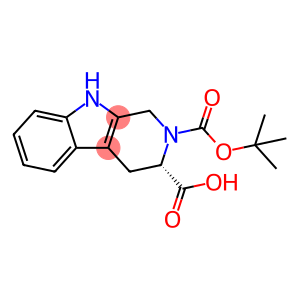
N-Boc-L-1,2,3,4-Tetrahydro-beta-carboline-3-carboxylic acid
CAS: 66863-43-2
Molecular Formula: C17H20N2O4
66863-43-2 - Names and Identifiers
66863-43-2 - Physico-chemical Properties
| Molecular Formula | C17H20N2O4 |
| Molar Mass | 316.35 |
| Density | 1.322±0.06 g/cm3(Predicted) |
| Melting Point | 286-291 °C |
| Boling Point | 525.6±50.0 °C(Predicted) |
| Flash Point | 271.7°C |
| Vapor Presure | 7.12E-12mmHg at 25°C |
| BRN | 691288 |
| pKa | 3.98±0.20(Predicted) |
| Storage Condition | 0-6°C |
66863-43-2 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| HS Code | 29339900 |
| Hazard Class | IRRITANT |
66863-43-2 - Introduction
N-Boc-L-1,2,3, acid(N-Boc-L-1,2,3, acid) is an organic compound. The following is a detailed description of its nature, use, formulation and safety information:
Nature:
- Molar Mass (molar mass):353.42g/mol
-Appearance: Colorless to slightly yellow solid
-Melting point: about 140-145°C
-Solubility: Soluble in common organic solvents (such as dimethyl sulfoxide, ethanol, dichloromethane)
-Chemical structure:
O
//
H H H H O
| | | | ||
H--C--C--C--C--C--C--C--O--OH
// || || || ||
-H R R H CH2 H
Use:
N-Boc-L-1,2,3, acid are usually used as raw materials for organic synthesis reactions. It can be used as a catalyst and intermediate in natural product synthesis or drug synthesis.
Preparation Method:
The preparation method of N-Boc-L-1,2,3, acid can be completed by the following steps:
1. Dissolve L-1 2,3, 4-tetrahydro-beta-carboline in dry dichloromethane.
2. Add excess t-butyl dicarbonyl imine to the reaction system, and add excess trimethyl boron oxide as an acid reagent.
3. When the reaction proceeds, the temperature of the reaction system is maintained at 0-5°C, and the reaction is allowed to proceed for 24 hours under stirring.
4. After the reaction is completed, the organic phase is washed with deionized water, and the solvent is evaporated to obtain the target product.
Safety Information:
N-Boc-L-1,2,3, acid safety information is currently less reported. However, as a chemical substance, use should follow appropriate laboratory safety procedures. It is recommended to wear personal protective equipment such as lab gloves and goggles to avoid exposure to the compound. In case of accidental swallowing or contact with skin and eyes, seek medical help immediately.
Nature:
- Molar Mass (molar mass):353.42g/mol
-Appearance: Colorless to slightly yellow solid
-Melting point: about 140-145°C
-Solubility: Soluble in common organic solvents (such as dimethyl sulfoxide, ethanol, dichloromethane)
-Chemical structure:
O
//
H H H H O
| | | | ||
H--C--C--C--C--C--C--C--O--OH
// || || || ||
-H R R H CH2 H
Use:
N-Boc-L-1,2,3, acid are usually used as raw materials for organic synthesis reactions. It can be used as a catalyst and intermediate in natural product synthesis or drug synthesis.
Preparation Method:
The preparation method of N-Boc-L-1,2,3, acid can be completed by the following steps:
1. Dissolve L-1 2,3, 4-tetrahydro-beta-carboline in dry dichloromethane.
2. Add excess t-butyl dicarbonyl imine to the reaction system, and add excess trimethyl boron oxide as an acid reagent.
3. When the reaction proceeds, the temperature of the reaction system is maintained at 0-5°C, and the reaction is allowed to proceed for 24 hours under stirring.
4. After the reaction is completed, the organic phase is washed with deionized water, and the solvent is evaporated to obtain the target product.
Safety Information:
N-Boc-L-1,2,3, acid safety information is currently less reported. However, as a chemical substance, use should follow appropriate laboratory safety procedures. It is recommended to wear personal protective equipment such as lab gloves and goggles to avoid exposure to the compound. In case of accidental swallowing or contact with skin and eyes, seek medical help immediately.
Last Update:2024-04-09 20:45:29
Supplier List
Product Name: BOC-L-1,2,3,4-TETRAHYDRONORHARMAN-3-CARBOXYLIC ACID Request for quotation
CAS: 66863-43-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 66863-43-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: Boc-L-1,2,3,4-Tetrahydronorharman-3-Carboxylic Acid Visit Supplier Webpage Request for quotation
CAS: 66863-43-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 66863-43-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: BOC-L-1,2,3,4-TETRAHYDRONORHARMAN-3-CARBOXYLIC ACID Request for quotation
CAS: 66863-43-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 66863-43-2
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: Boc-L-1,2,3,4-Tetrahydronorharman-3-Carboxylic Acid Visit Supplier Webpage Request for quotation
CAS: 66863-43-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 66863-43-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History

