65-31-6
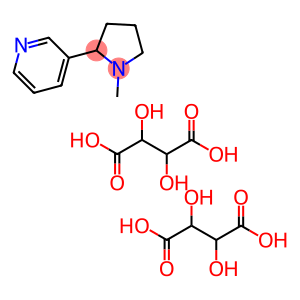
nicotine dihydrogen ditartrate
CAS: 65-31-6
Molecular Formula: C18H26N2O12
65-31-6 - Names and Identifiers
| Name | nicotine dihydrogen ditartrate |
| Synonyms | Nicotine tartrate (-)-Nicotine ditatrate (-)-Nicotine dirartate Nicotine Bitartrate anhydrous nicotine dihydrogen ditartrate 3-[(2S)-1-Methylpyrrolidin-2-yl]pyridine ditartrate (S)-(-)-1-Methyl-2-(3-pyridyl)pyrrolidine(+)-ditartratesalt (-)-1-Methyl-2-(3-pyridyl)pyrrolidine (+)-bitartrate salt 3-[(2S)-1-methylpyrrolidin-2-yl]pyridine 2,3-dihydroxybutanedioate (salt) 3-[(2S)-1-methylpyrrolidin-2-yl]pyridine bis(2,3-dihydroxybutanedioate) (salt) |
| CAS | 65-31-6 |
| EINECS | 200-607-2 |
| InChI | InChI=1/C10H14N2.C4H6O6/c1-12-7-3-5-10(12)9-4-2-6-11-8-9;5-1(3(7)8)2(6)4(9)10/h2,4,6,8,10H,3,5,7H2,1H3;1-2,5-6H,(H,7,8)(H,9,10)/t10-;/m0./s1 |
| InChIKey | RFEJUZJILGIRHQ-UHFFFAOYSA-N |
65-31-6 - Physico-chemical Properties
| Molecular Formula | C18H26N2O12 |
| Molar Mass | 462.41 |
| Melting Point | 90° |
| Boling Point | 244.4°C at 760 mmHg |
| Specific Rotation(α) | D20 +26° (c = 10) |
| Flash Point | 101.7°C |
| Water Solubility | 1000000 mg l-1 (e) |
| Solubility | H2O: 50mg/mL |
| Vapor Presure | 0.0303mmHg at 25°C |
| Appearance | powder |
| Color | white to off-white |
| Storage Condition | Desiccate at RT |
| Physical and Chemical Properties | Used in smoking cessation treatment. |
65-31-6 - Risk and Safety
| Risk Codes | R26/27/28 - Very toxic by inhalation, in contact with skin and if swallowed. R51/53 - Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S13 - Keep away from food, drink and animal foodstuffs. S28 - After contact with skin, wash immediately with plenty of soap-suds. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 1659 6.1/PG 2 |
| WGK Germany | 2 |
| RTECS | QT0350000 |
| Hazard Class | 6.1(a) |
| Packing Group | II |
65-31-6 - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Overview | nicotine common name nicotine, belonging to the pyridine derivatives alkaloids, with a certain degree of alkalinity, and acid reaction to produce amine salts. Therefore, nicotine salt (nicotine salt) is essentially an amine salt, and nicotine salt (nicotine salt) is much less irritating to the mucous membrane of the mouth or throat than nicotine itself. |
| human metabolism | nicotine salt (nicotine salt) will be decomposed into nicotine and the corresponding acid after entering the human body, nicotine salt of the same quality (nicotine salt) and nicotine, in fact, the amount of nicotine into the human body is less, nicotine salt (nicotine salt) in the weight of the acid, therefore, nicotine salt (nicotine salt) can generally be added to 50mg, or even nearly 100mg, but in fact, the amount of real nicotine may not even be generally, nicotine salt (nicotine salt) in cigarette oil it's made with citric acid and tartaric acid. |
| category | toxic substances |
| toxicity grade | high toxicity |
| Acute toxicity | oral-rat LD50: 65 mg/kg; Oral-mouse LD50: 65 mg/kg |
| storage and transportation characteristics | The warehouse is ventilated and dried at low temperature; It is stored and transported separately from food raw materials |
| extinguishing agent | sand, water, foam, carbon dioxide, dry powder |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 19:55:45
Supplier List
Product Name: nicotine dihydrogen ditartrate Request for quotation
CAS: 65-31-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 65-31-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
Product Name: nicotine dihydrogen ditartrate Request for quotation
CAS: 65-31-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 65-31-6
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
View History
