646-97-9
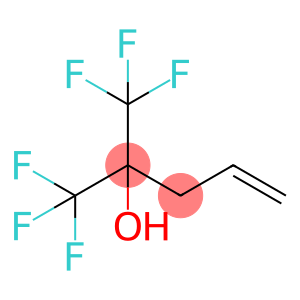
2-Allylhexafluoroisopropanol
CAS: 646-97-9
Molecular Formula: C6H6F6O
646-97-9 - Names and Identifiers
| Name | 2-Allylhexafluoroisopropanol |
| Synonyms | BTHB ALLYLHEXAFLUOROISO-PROPANOL 2-ALLYLHEXAFLUOROISOPROPANOL 2-Allylhexafluoroisopropanol 2-[(2-chloro-6-fluorobenzyl)sulfanyl]aniline 1,1,1-TRIFLUORO-2-TRIFLUOROMETHYL-PENT-4-ENE-2-OL 1,1,1-TRIFLUORO-2-(TRIFLUOROMETHYL)PENT-4-EN-2-OL 1,1,1-Trifluoro-2-(trifluoromethyl)pent-4-en-2-ol 1,1,1-Trifluoro-2-(trifluoromethyl)-4-penten-2-ol |
| CAS | 646-97-9 |
| EINECS | 613-651-6 |
| InChI | InChI=1/C13H11ClFNS/c14-10-4-3-5-11(15)9(10)8-17-13-7-2-1-6-12(13)16/h1-7H,8,16H2 |
646-97-9 - Physico-chemical Properties
| Molecular Formula | C6H6F6O |
| Molar Mass | 208.1 |
| Density | 1.36g/ml |
| Boling Point | 97 °C |
| Flash Point | 183.4°C |
| Vapor Presure | 5.8E-06mmHg at 25°C |
| Appearance | clear liquid |
| Color | Colorless to Light yellow |
| pKa | 9.62±0.29(Predicted) |
| Storage Condition | Room Temprature |
| Refractive Index | 1.3320 to 1.3360 |
646-97-9 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | R10 - Flammable R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S16 - Keep away from sources of ignition. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| UN IDs | 1987 |
| Hazard Note | Irritant |
| Hazard Class | IRRITANT, LACHRYMATO |
| Packing Group | III |
646-97-9 - Introduction
It is an organic compound with the chemical formula C6H7F6O. The following is a description of its nature, use, preparation and safety information:
Nature:
-Appearance: colorless liquid.
-Density: Its density is about 1.4 g/mL.
-Boiling point and melting point: Its boiling point is about 102-103°C, and its melting point is about -30°C.
-Solubility: Soluble in many polar organic solvents, such as ethanol, acetone and dimethylformamide.
Use:
-Chemical reaction reagent: It can be used as a reagent in chemical reactions, for example, as a reducing agent for carbonyl compounds, or as an intermediate for the construction of organic molecules.
-Catalyst: It can also be used as a catalyst for certain chemical reactions, such as the reaction between enolone and amino compounds.
-Surfactant: Due to its unique chemical structure, it can also be used as a surfactant to improve wetting properties.
Preparation Method:
-It is usually synthesized by the following steps:
1. First, hexafluoroisopropanol is reacted with bromopropene to generate 2-bromoisopropylhexafluoroisopropanol under the catalysis of alkali.
2. Then react 2-bromoisopropylhexafluoroisopropanol with a base, such as potassium hydroxide or sodium carbonate, to obtain the final product.
Safety Information:
-the use and storage of the need to be careful, should pay attention to the following safety matters:
-Use with appropriate protective gloves and eye protection. Avoid exposure to skin and eyes.
-avoid inhalation of its vapor, try to use in a well-ventilated place.
-Ensure good operational controls and precautions when conducting experiments or industrial applications.
Please note that the above information is for reference only. When using chemical substances, you should carefully read and follow the relevant safety guidelines and operating procedures.
Nature:
-Appearance: colorless liquid.
-Density: Its density is about 1.4 g/mL.
-Boiling point and melting point: Its boiling point is about 102-103°C, and its melting point is about -30°C.
-Solubility: Soluble in many polar organic solvents, such as ethanol, acetone and dimethylformamide.
Use:
-Chemical reaction reagent: It can be used as a reagent in chemical reactions, for example, as a reducing agent for carbonyl compounds, or as an intermediate for the construction of organic molecules.
-Catalyst: It can also be used as a catalyst for certain chemical reactions, such as the reaction between enolone and amino compounds.
-Surfactant: Due to its unique chemical structure, it can also be used as a surfactant to improve wetting properties.
Preparation Method:
-It is usually synthesized by the following steps:
1. First, hexafluoroisopropanol is reacted with bromopropene to generate 2-bromoisopropylhexafluoroisopropanol under the catalysis of alkali.
2. Then react 2-bromoisopropylhexafluoroisopropanol with a base, such as potassium hydroxide or sodium carbonate, to obtain the final product.
Safety Information:
-the use and storage of the need to be careful, should pay attention to the following safety matters:
-Use with appropriate protective gloves and eye protection. Avoid exposure to skin and eyes.
-avoid inhalation of its vapor, try to use in a well-ventilated place.
-Ensure good operational controls and precautions when conducting experiments or industrial applications.
Please note that the above information is for reference only. When using chemical substances, you should carefully read and follow the relevant safety guidelines and operating procedures.
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: 1,1,1-Trifluoro-2-(trifluoromethyl)-4-penten-2-ol Visit Supplier Webpage Request for quotationCAS: 646-97-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,1,1-TRIFLUORO-2-(TRIFLUOROMETHYL)PENT-4-EN-2-OL Request for quotation
CAS: 646-97-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 646-97-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 2-Allylhexafluoroisopropanol Visit Supplier Webpage Request for quotation
CAS: 646-97-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 646-97-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 1,1,1-Trifluoro-2-(trifluoromethyl)-4-penten-2-ol Visit Supplier Webpage Request for quotationCAS: 646-97-9
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 1,1,1-TRIFLUORO-2-(TRIFLUOROMETHYL)PENT-4-EN-2-OL Request for quotation
CAS: 646-97-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 646-97-9
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 2-Allylhexafluoroisopropanol Visit Supplier Webpage Request for quotation
CAS: 646-97-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 646-97-9
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


