6141-13-5
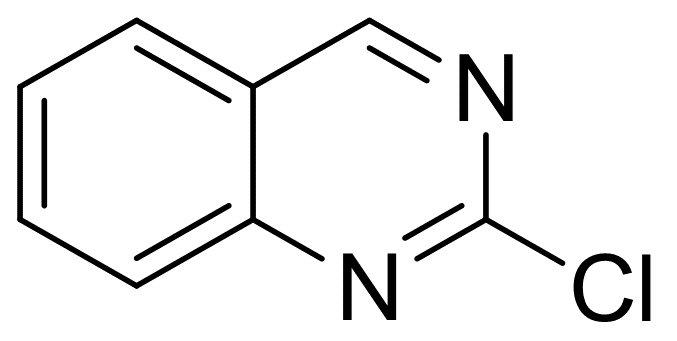
2-Chloroquinazoline
CAS: 6141-13-5
Molecular Formula: C8H5ClN2
6141-13-5 - Names and Identifiers
| Name | 2-Chloroquinazoline |
| Synonyms | 2-chloroquinazoline 2-Chloroquinazoline Quinazoline, 2-chloro- 2-Chloroquinazoline(WX636001) |
| CAS | 6141-13-5 |
| InChI | InChI=1/C8H5ClN2/c9-8-10-5-6-3-1-2-4-7(6)11-8/h1-5H |
6141-13-5 - Physico-chemical Properties
| Molecular Formula | C8H5ClN2 |
| Molar Mass | 164.59 |
| Density | 1.349±0.06 g/cm3(Predicted) |
| Melting Point | 108 °C(Solv: ligroine (8032-32-4)) |
| Boling Point | 250.0±13.0 °C(Predicted) |
| pKa | 0.22±0.30(Predicted) |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
| MDL | MFCD09959715 |
6141-13-5 - Risk and Safety
| Risk Codes | 41 - Risk of serious damage to eyes |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S39 - Wear eye / face protection. |
6141-13-5 - Reference Information
| Application | 2-chloroquinazoline is an organic intermediate that can be made of benzoylene Urea is used as raw material to prepare 2,4-dichloroquinazoline, and then dechlorinated to obtain 2-chloroquinazoline. |
| preparation | 2,4-dichloroquinazoline (IIIA) 2.0 grams of benzoylurea and 0.6mL of N,N-dimethylaniline are put into 6mL of phosphorus oxychloride, refluxing for 7 hours, cooling to room temperature, and depressurizing most of phosphorus oxychloride, the remaining reaction liquid is transferred to ice water, stirred to generate a large amount of khaki solid, filter, filter cake is washed with distilled water, and after drying, 1.6 grams of yellow-green solid (IIIA) is obtained, and the yield is 65%. Mp118-119 ℃;1H-NMR(400Hz,CDCl3),δ:7.75-7.79(m,1H),8.02-8.03(m,2H),8.29(d,J = 8.4Hz,1H).2-chloroquinazoline (IIIB) will be 0.5 grams of 2,4-dichloroquinazoline (IIIA), 10mL of saturated saline containing 9% NH4OH and 0.5g of activated zinc powder were fed into a eggplant flask containing 10mL of dichloromethane and refluxed for 4 hours. Cooling, suction filtration to remove zinc powder, filtrate under reduced pressure to evaporate solvent, extraction with ethyl acetate, the resulting organic layer is dried, filtered, reduced pressure to evaporate solvent, residue column chromatography separation of 0.2 grams of 2-chloroquinazoline (IIIB), the yield is 40%. |
Last Update:2024-04-09 21:11:58
Supplier List
Product Name: 2-Chloroquinazoline Request for quotation
CAS: 6141-13-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6141-13-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-Chloroquinazoline Visit Supplier Webpage Request for quotationCAS: 6141-13-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Chloroquinazoline Visit Supplier Webpage Request for quotation
CAS: 6141-13-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6141-13-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2-Chloroquinazoline Request for quotation
CAS: 6141-13-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6141-13-5
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-Chloroquinazoline Visit Supplier Webpage Request for quotationCAS: 6141-13-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-Chloroquinazoline Visit Supplier Webpage Request for quotation
CAS: 6141-13-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6141-13-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


