6000-50-6
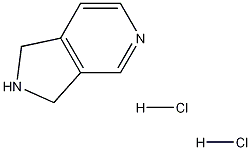
2,3-Dihydro-1H-Pyrrolo[3,4-C]Pyridine dihydrochloride
CAS: 6000-50-6
Molecular Formula: C7H10Cl2N2
6000-50-6 - Names and Identifiers
6000-50-6 - Physico-chemical Properties
| Molecular Formula | C7H10Cl2N2 |
| Molar Mass | 193.0737 |
| Boling Point | 286.3°C at 760 mmHg |
| Flash Point | 126.9°C |
| Vapor Presure | 0.00203mmHg at 25°C |
| Storage Condition | under inert gas (nitrogen or Argon) at 2-8°C |
6000-50-6 - Reference Information
| application | 2, 3-dihydro-1H-pyrrole [3,4-C] pyridine dihydrochloride can be used as an intermediate in pharmaceutical synthesis, which can be prepared from propargylamine and ethyl chloroformate through four-step reaction. There are reports that it can be used to prepare an A2A receptor antagonist. |
| preparation | a)N-propargyl urethane take 115 grams (2.1 mol) propargyl amine dissolved in 1 liter of toluene, add 91 grams of sodium hydroxide dissolved in 400 ml of water, and add 239 grams (2.2 mol) of ethyl chloroformate drop by drop at 10 ℃. The mixture was stirred at room temperature for three hours to separate the organic phase, the aqueous phase was extracted with toluene, and the organic solution was dried with magnesium sulfate, concentrated and distilled. The yield is 221g (83% of theoretical value) and the boiling point is 101°C / 20 mbar. B) 2-(N-ethoxycarbonyl-N-propargyl aminomethyl) pyrimidine 11.5g(91 mmol)N-propargyl urethane was dissolved in 90 ml toluene, 20.3g KOH powder, 0.5g triethylbenzylammonium chloride and 13.5g(104 mmol) 2-chloromethylpyrimidine (DOS 2 932 643) were added. The mixture was stirred overnight at room temperature, the salt was pumped out, the filtrate was washed with a saturated sodium chloride solution, dried with magnesium sulfate, concentrated and distilled. The yield is 10.4g (51% of theoretical value) and the boiling point is 130°C/0.35mbar. c) Ethyl 5, 7-dihydro-6H-pyrrolo [3,4-b] pyridine-6-carboxylate refluxing 7.7g(35mmol) of 2-(N-ethoxycarbonyl-N-propargylaminomethyl) pyrimidine in 150ml xylene for 40 h, concentrating it and recrystallizing the residue from petroleum ether. The yield is 5.5g (81.7% of theoretical value) and the melting point is 77-79°C. d)2, 3-dihydro-1H-pyrrole [3,4-C] pyridine dihydrochloride dihydrochloride 8.5g(44mmol) of 5, 7-dihydro-1H-pyrrolo [3,4-b] pyridine -6-carboxylate ethyl ester was heated and refluxed overnight in 90ml of concentrated hydrochloric acid. Concentrate the solution, the residue is stirred with acetone, the salt is pumped out, and dried in air. The yield is 7.5 grams (88% of theoretical value). |
Last Update:2024-04-09 15:16:35
Supplier List
Product Name: 2,3-Dihydro-1H-Pyrrolo[3,4-C]Pyridine dihydrochloride Request for quotation
CAS: 6000-50-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6000-50-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2,3-Dihydro-1H-pyrrolo[3,4-c]pyridine Dihydrochloride Visit Supplier Webpage Request for quotationCAS: 6000-50-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3-Dihydro-1H-Pyrrolo[3,4-C]Pyridine dihydrochloride Visit Supplier Webpage Request for quotation
CAS: 6000-50-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6000-50-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: 2,3-Dihydro-1H-Pyrrolo[3,4-C]Pyridine dihydrochloride Request for quotation
CAS: 6000-50-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6000-50-6
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2,3-Dihydro-1H-pyrrolo[3,4-c]pyridine Dihydrochloride Visit Supplier Webpage Request for quotationCAS: 6000-50-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2,3-Dihydro-1H-Pyrrolo[3,4-C]Pyridine dihydrochloride Visit Supplier Webpage Request for quotation
CAS: 6000-50-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6000-50-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


