59-97-2
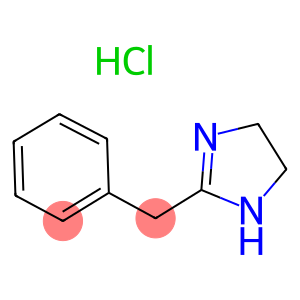
Tolazoline hydrochloride
CAS: 59-97-2
Molecular Formula: C10H13ClN2
59-97-2 - Names and Identifiers
| Name | Tolazoline hydrochloride |
| Synonyms | Tolazoline HCl TIMTEC-BB SBB000360 2-BENZYL-2-IMIDAZOLINE Tolazoline hydrochloride BENZIDAZOL HYDROCHLORIDE BENZAZOLINE HYDROCHLORIDE 2-BENZYL-4,5-IMIDAZOLINE HCL 2-BENZYL-2-IMIDAZOLINE CHLORHYDRATE 2-BENZYL-2-IMIDAZOLINE HYDROCHLORIDE 2-BENZYL-2-IMINAZOLINE HYDROCHLORIDE 2-Benzyl-2-imidazoline hydrochloride |
| CAS | 59-97-2 |
| EINECS | 200-447-3 |
| InChI | InChI=1/C10H12N2.ClH/c1-2-4-9(5-3-1)8-10-11-6-7-12-10;/h1-5H,6-8H2,(H,11,12);1H |
59-97-2 - Physico-chemical Properties
| Molecular Formula | C10H13ClN2 |
| Molar Mass | 196.68 |
| Melting Point | 172-176 °C |
| Boling Point | 338.2°C at 760 mmHg |
| Flash Point | 158.3°C |
| Solubility | DMSO (Slightly, Heated), Methanol (Slightly) |
| Vapor Presure | 0.000196mmHg at 25°C |
| Appearance | Crystalline Powder |
| Color | White to almost white |
| Merck | 14,9506 |
| Storage Condition | Inert atmosphere,Room Temperature |
| Stability | Hygroscopic |
| Sensitive | Hygroscopic |
| MDL | MFCD00012693 |
| Use | Used as vasodilator |
| In vitro study | Tolazoline is a pulmonary vasodilator used to attenuate pulmonary vascular resistance (PVR) in neonates with persistent pulmonary hypertension (PPHN). Tolazoline has moderate alpha-adrenergic blocking activity and histamine agonist activity. Tolazoline is commonly used to reduce pulmonary artery pressure and vascular resistance. For all the spasmogenic drugs investigated, Tolazoline did not have a broad range of effects as did SNP. However, in the human radial artery, it may be effective in counteracting alpha-adrenergic receptor-mediated vasospasm. |
59-97-2 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | 22 - Harmful if swallowed |
| Safety Description | 22 - Do not breathe dust. |
| UN IDs | 2811 |
| TSCA | Yes |
| HS Code | 29332900 |
| Toxicity | LD50 orl-rat: 1200 mg/kg NIIRDN 6,511,82 |
59-97-2 - Standard
Authoritative Data Verified Data
This product is 4, 5-dihydro-2-benzyl-1h-imidazole hydrochloride. The content of C10H12N2 • HCl shall not be less than 98.5% calculated on a dry basis.
59-97-2 - Trait
Authoritative Data Verified Data
- This product is white or off-white crystalline powder. This product of the aqueous solution of litmus paper microscopic acidic reaction.
- This product is soluble in water, soluble in ethanol or chloroform, insoluble in ether.
melting point
The melting point of this product (General 0612) is 172~176°C.
59-97-2 - Differential diagnosis
Authoritative Data Verified Data
- take about 10mg of this product, add 1 ml of water to dissolve, add several drops of ammonium chromium thiocyanate test solution, and generate pink precipitate.
- take about 10mg of this product, add 1 ml of water to dissolve, add trinitrophenol test solution to no longer produce precipitation, filter, precipitate wash, dry, and determine according to law, the melting point is 144 to 149 ° C. (General 0612).
- The infrared absorption spectrum of this product should be consistent with that of the control (Spectrum set 1193).
- the aqueous solution of this product was chloride identification (1) of the reaction (General 0301).
59-97-2 - Exam
Authoritative Data Verified Data
loss on drying
take this product, dry to constant weight at 105°C, weight loss shall not exceed 0.5% (General rule 0831).
ignition residue
not more than 0.1% (General rule 0841).
59-97-2 - Content determination
Authoritative Data Verified Data
take this product about 0.15g, precision weighing, add glacial acetic acid 20ml dissolved, add mercury acetate test solution 5ml and crystal violet indicator solution 1 drop, with perchloric acid titration solution (0.1 mol/L) titration to the solution blue-green, and the titration results were corrected with a blank test. Per 1 ml of perchloric acid titration solution (0.1 mol/L) corresponds to 19.67mg of C10H12N2-hc1.
59-97-2 - Category
Authoritative Data Verified Data
a adrenergic receptor blockers.
59-97-2 - Storage
Authoritative Data Verified Data
light shielding, sealed storage.
59-97-2 - Tolazolin hydrochloride tablets
Authoritative Data Verified Data
This product shall contain tolazoline hydrochloride (C10H12N2 • HCl) in an amount of 93.0% to 107.0% of the labeled amount.
trait
This product is a sugar-coated tablet, which is white after removing the coating.
identification
Take 8 tablets of this product, grind, extract with ethanol for 3 times, 10ml each time, heat, filter, combine filtrate to dry, add water 10ml, dissolve, filter, filtrate to dry, the following tests were performed for residual ulcers.
- about 10mg of the residue was taken, and 1 ml of water was added to dissolve tolazoline hydrochloride, and several drops of a test solution of ammonium chromate thiocyanate were added to produce a pink precipitate.
- the aqueous solution of the residue showed the reaction of chloride identification (1) (General 0301).
examination
should be in accordance with the relevant provisions under The tablet item (General rule 0101).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica as filler; Methanol-0.068% phosphoric acid solution [(50:50), the pH value was adjusted to 3.0 with ammonia solution. The detection wavelength was 230nm. The number of theoretical plates shall not be less than 1500 based on the calculation of tazolin peak.
- determination of 20 tablets of this product, precision weighing, fine grinding, precision weighing an appropriate amount (about 50mg equivalent to tolazoline hydrochloride), put in a 100ml measuring flask, add an appropriate amount of mobile phase, ultrasonic dissolution of tolazoline hydrochloride, dilute to scale with mobile phase, shake well, filter, Take 5ml of continued filtrate precisely, put it in 50ml measuring flask, dilute to scale with mobile phase, shake well, accurately measure 20ul, inject human liquid chromatograph, record the chromatogram; Take another reference substance of tolazoline hydrochloride, the mobile phase was added to dissolve and quantitatively dilute to prepare a solution containing about 50ug per 1 ml, which was determined by the same method. According to the external standard method to calculate the peak area, that is.
category storage
Same as tolazoline hydrochloride.
specification
25mg
59-97-2 - Tolazolin hydrochloride injection
Authoritative Data Verified Data
This product is a sterile aqueous solution of tolazoline hydrochloride. Tzozoline hydrochloride CC10H12N2 • HC1) shall be between 95.0% and 105.0% of the label amount.
trait
This product is a clear colorless liquid.
identification
- take 1ml of this product, add a few drops of chromium ammonium thiocyanate test solution, which will produce pink precipitate.
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- This product chloride identification (1) of the reaction (General 0301).
examination
- the pH value should be 4.5 to 6.5 (General 0631).
- Related substances take this product and dilute it with methanol-water (1:2) to prepare a solution containing 0.5mg of turazolin hydrochloride per 1 ml as a test solution; take 1 ml accurately, put it in a 100ml measuring flask, dilute it to the scale with methanol-water (1:2), and shake it well to serve as a control solution. According to the chromatographic conditions under the content determination item, 20 u1 of each of the test solution and the control solution are accurately measured and injected into the human liquid chromatograph respectively, and the chromatogram is recorded to 4 times of the retention time of the main component chromatographic peak. If there are impurity peaks in the chromatogram of the test solution, the sum of each impurity peak area shall not be greater than the main peak area of the control solution (1.0%).
- the bacterial endotoxin of this product is taken and checked according to law (General rule 1143). The amount of endotoxin contained in every 1 mg of tolazolin hydrochloride should be less than 0.80EU.
- others should comply with the relevant provisions under injection (General 0102).
Content determination
- determined by HPLC (General 0512)
- chromatographic conditions and system suitability test using eighteen alkyl silane bonded silica as filler; Methanol-0.068% phosphoric acid solution [(50:50), the pH value was adjusted to 3.0 with ammonia solution. The detection wavelength was 230nm. The number of theoretical plates shall not be less than 1500 based on the calculation of turazolin peak, and the separation degree between turazolin peak and adjacent impurity peaks shall be in accordance with the regulations.
- precision measurement: take an appropriate amount of this product (about 50mg equivalent to tolazoline hydrochloride), put it in a 100ml measuring flask, dilute it to the scale with mobile phase, shake it well, and take 5ml of precision measurement, put it in a 50ml measuring flask, dilute to the scale with mobile phase, shake well, use 20ul as test solution, inject human liquid chromatograph, record chromatogram; another reference substance of tolazolin hydrochloride was precision weighed, dissolved and quantitatively diluted with mobile phase to prepare a solution containing 0.05mg per 1 ml, which was determined by the same method. According to the external standard method to calculate the peak area, that is.
category
Same as tolazoline hydrochloride.
specification
lml:25mg
storage
protected from light and sealed.
Supplier List
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 59-97-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 59-97-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 59-97-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 59-97-2
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 59-97-2
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 59-97-2
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025



