57044-25-4
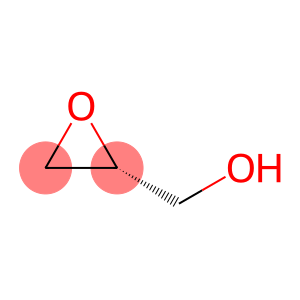
R(+)-glycidol
CAS: 57044-25-4
Molecular Formula: C3H6O2
57044-25-4 - Names and Identifiers
| Name | R(+)-glycidol |
| Synonyms | EOS-60676 (R)-Glycidol (R)-GLYCIDOL R(+)-glycidol R-(+)-Glycidol (R)-(+)-GLYCIDOL (R)-(+)-Glycidol (r)-oxiranemethano (R)-Oxiranemethanol OXIRANEMETHANOL, (2R)- R(+)-OXIRANE-2-METHANOL (R)-(+)-2,3-Epoxy-1-Propanol (R)-(+)-2,3-EPOXY-1-PROPANOL |
| CAS | 57044-25-4 |
| EINECS | 404-660-4 |
| InChI | InChI=1/C3H6O2/c4-1-3-2-5-3/h3-4H,1-2H2 |
| InChIKey | CTKINSOISVBQLD-GSVOUGTGSA-N |
57044-25-4 - Physico-chemical Properties
| Molecular Formula | C3H6O2 |
| Molar Mass | 74.08 |
| Density | 1.116g/mLat 20°C(lit.) |
| Boling Point | 56-57°C11mm Hg(lit.) |
| Flash Point | 178°F |
| Water Solubility | Completely miscible in water |
| Appearance | neat |
| Color | Colorless to Light yellow |
| BRN | 79782 |
| pKa | 14.62±0.10(Predicted) |
| Storage Condition | -20°C |
| Refractive Index | n20/D 1.43(lit.) |
| Physical and Chemical Properties | Colorless to light yellow liquid, stored in a closed container at room temperature. Boiling point 163-164°C. |
| Use | For the synthesis of a variety of Chiral Pharmaceutical Intermediates |
57044-25-4 - Risk and Safety
| Risk Codes | R45 - May cause cancer R60 - May impair fertility R2 - Risk of explosion by shock, friction, fire or other sources of ignition R21/22 - Harmful in contact with skin and if swallowed. R23 - Toxic by inhalation R34 - Causes burns R68 - Possible risk of irreversible effects R41 - Risk of serious damage to eyes R37/38 - Irritating to respiratory system and skin. |
| Safety Description | S53 - Avoid exposure - obtain special instructions before use. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| UN IDs | UN 2922 8/PG 2 |
| WGK Germany | 3 |
| RTECS | RR0508000 |
| FLUKA BRAND F CODES | 10-21 |
| HS Code | 29109000 |
| Hazard Class | 6.1 |
| Packing Group | Ⅲ |
57044-25-4 - Reference Information
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| application | (R)-glycidyl can be used to synthesize a variety of chiral pharmaceutical intermediates, and can be used in laboratory research and development processes and chemical production processes. |
| Synthesis method | In the stirred state, slowly add NaOH(36.19g, 0.905 moles) to the chlordiol (100.0g, 0.905 moles) and ethanol (200 ml) solution at 0-5 degrees, and then stir the reaction mixture at 0-5 degrees for 2 hours. After the reaction is over, the reaction mixture is filtered to remove the insoluble solid precipitate, and the obtained filtrate is vacuum concentrated at 40 degrees to obtain the target product (R)-glycidyl (note that the boiling point of (R)-glycidyl is low, and low temperature treatment is required when the post-treatment is vacuum concentrated). Fig. (R)-synthetic route of glycidyl |
| traits | (R)-glycidyl is a colorless to light yellow liquid. |
| Use | Used to construct chiral structural units of epoxy vinyl iodide intermediates in the synthesis of furanocembrane (a marine natural product). for the synthesis of a variety of chiral pharmaceutical intermediates |
Last Update:2024-04-09 20:13:35
Supplier List
Product Name: (R)-(+)-Glycidol Request for quotation
CAS: 57044-25-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 57044-25-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: R-(+)-Glycidol Visit Supplier Webpage Request for quotationCAS: 57044-25-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (R)-(+)-Glycidol Request for quotation
CAS: 57044-25-4
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 57044-25-4
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
Product Name: (R)-(+)-Glycidol Request for quotation
CAS: 57044-25-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 57044-25-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: R-(+)-Glycidol Visit Supplier Webpage Request for quotationCAS: 57044-25-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (R)-(+)-Glycidol Request for quotation
CAS: 57044-25-4
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
CAS: 57044-25-4
Tel: 0086-13605431940
Email: xuli678323@126.com
Mobile: +86-13605431940
QQ: 56860987
Wechat: 13605431940
WhatsApp: 13605431940
View History


