5636-65-7
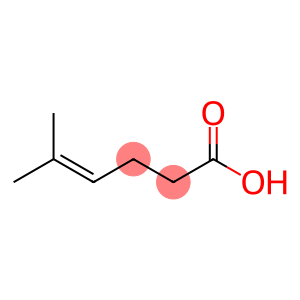
5-methylhex-4-enoic acid
CAS: 5636-65-7
Molecular Formula: C7H12O2
5636-65-7 - Names and Identifiers
| Name | 5-methylhex-4-enoic acid |
| Synonyms | 5-METHYL-4-HEXENOICACID 5-Methylhex-4-enoic acid 5-methylhex-4-enoic acid 5-METHYL-4-HEXENOIC ACID 4-hexenoic acid, 5-methyl- 4-Hexenoic acid, 5-methyl- (3E)-5-methylhex-3-enoic acid |
| CAS | 5636-65-7 |
| InChI | InChI=1/C7H12O2/c1-6(2)4-3-5-7(8)9/h4H,3,5H2,1-2H3,(H,8,9) |
5636-65-7 - Physico-chemical Properties
| Molecular Formula | C7H12O2 |
| Molar Mass | 128.17 |
| Density | 0.9862 |
| Melting Point | -28°C |
| Boling Point | 237.28°C (estimate) |
| Flash Point | 117.993°C |
| Vapor Presure | 0.052mmHg at 25°C |
| pKa | pK1:4.8 (25°C) |
| Refractive Index | 1.4504 |
5636-65-7 - Introduction
5-methylhex-4-enoic acid(5-methylhex-4-enoic acid) is an organic compound with the molecular formula C7H12O2. The following is an introduction to its nature, use, preparation and safety information:
Nature:
5-methylhex-4-enoic acid is a colorless to pale yellow liquid with a special smell. It has a density of 0.930g/cm³, a melting point of -10°C and a boiling point of 203-205°C. Its solubility is good, soluble in water and common organic solvents.
Use:
5-methylhex-4-enoic acid is mainly used in the field of organic synthesis. It can be used as a raw material or intermediate in organic synthesis reactions, and is commonly used in the synthesis of biologically active compounds, fragrances, drugs and pesticides.
Preparation Method:
There are two main methods for preparing 5-methylhex-4-enoic acid:
1. Through the reaction of hexanedione and methyl ether diborate in the presence of potassium carbonate, 5-methyl -4-hexene -2,3-alcohol acid ester is generated, and then the target product is obtained by acid catalyzed heating hydrolysis.
2. through hexanal and isobutyl magnesium halide Michael addition reaction to generate 5-methyl -4-hexenol, and then through the oxidation reaction to produce the target product.
Safety Information:
5-methylhex-4-enoic acid is an organic compound, and attention should be paid to its protective measures. It is irritating to the eyes and skin, avoid direct contact. Wear appropriate protective gloves, goggles and protective clothing during operation. In case of accidental contact, rinse the affected area immediately with plenty of water and seek medical help. During storage and use, keep away from fire and oxidant and keep well ventilated. Avoid contact with oxidizing substances, strong acids and strong bases to prevent dangerous reactions.
Nature:
5-methylhex-4-enoic acid is a colorless to pale yellow liquid with a special smell. It has a density of 0.930g/cm³, a melting point of -10°C and a boiling point of 203-205°C. Its solubility is good, soluble in water and common organic solvents.
Use:
5-methylhex-4-enoic acid is mainly used in the field of organic synthesis. It can be used as a raw material or intermediate in organic synthesis reactions, and is commonly used in the synthesis of biologically active compounds, fragrances, drugs and pesticides.
Preparation Method:
There are two main methods for preparing 5-methylhex-4-enoic acid:
1. Through the reaction of hexanedione and methyl ether diborate in the presence of potassium carbonate, 5-methyl -4-hexene -2,3-alcohol acid ester is generated, and then the target product is obtained by acid catalyzed heating hydrolysis.
2. through hexanal and isobutyl magnesium halide Michael addition reaction to generate 5-methyl -4-hexenol, and then through the oxidation reaction to produce the target product.
Safety Information:
5-methylhex-4-enoic acid is an organic compound, and attention should be paid to its protective measures. It is irritating to the eyes and skin, avoid direct contact. Wear appropriate protective gloves, goggles and protective clothing during operation. In case of accidental contact, rinse the affected area immediately with plenty of water and seek medical help. During storage and use, keep away from fire and oxidant and keep well ventilated. Avoid contact with oxidizing substances, strong acids and strong bases to prevent dangerous reactions.
Last Update:2024-04-09 15:17:58
Supplier List
Product Name: 5-METHYL-4-HEXENOIC ACID Request for quotation
CAS: 5636-65-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5636-65-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 5-METHYL-4-HEXENOIC ACID Request for quotation
CAS: 5636-65-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5636-65-7
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
View History
