56180-94-0
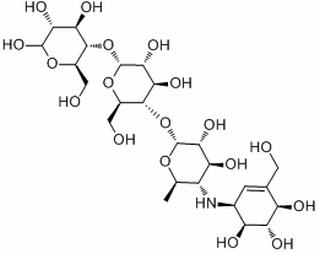
acarbose
CAS: 56180-94-0
Molecular Formula: C25H43NO18
56180-94-0 - Names and Identifiers
| Name | acarbose |
| Synonyms | acarbose Glucobay Acarbose Acarbose API Acarbose Hydrate Acarbose (Acarbosum) AMylostatin J, Bay g 5421, alpha-GHI (2R,3R,4R,5S,6R)-5-((2R,3R,4R,5S,6R)-5- O-4,6-Dideoxy-4-[[[1S-(1α,4α,5β,6α)]-4,5,6-trihydroxy-3-(hydroxyMethyl)-2-cyclohexen-1-yl]aMino]-α-D-glucopyranosyl-(1-4)-O-α-D-glucopyranosyl-(1-4)-D-glucose |
| CAS | 56180-94-0 |
| EINECS | 260-030-7 |
| InChI | InChI=1/C25H43NO18/c1-6-11(26-8-2-7(3-27)12(30)15(33)13(8)31)14(32)19(37)24(40-6)43-22-10(5-29)42-25(20(38)17(22)35)44-21-9(4-28)41-23(39)18(36)16(21)34/h2,6,8-39H,3-5H2,1H3 |
| InChIKey | XUFXOAAUWZOOIT-JMPDRRIHSA-N |
56180-94-0 - Physico-chemical Properties
| Molecular Formula | C25H43NO18 |
| Molar Mass | 645.61 |
| Density | 1.4278 (rough estimate) |
| Melting Point | 165-170°C |
| Boling Point | 675.05°C (rough estimate) |
| Specific Rotation(α) | D18 +165° (c = 0.4 in water) |
| Flash Point | 541.4°C |
| Water Solubility | Soluble in water. |
| Solubility | Soluble in water: 50mg/ml |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | White or white-like powder |
| Color | White to Off-White |
| Merck | 14,18 |
| pKa | 12.39±0.20(Predicted) |
| Storage Condition | 2-8°C |
| Stability | Hygroscopic |
| Refractive Index | 1.6000 (estimate) |
| MDL | MFCD00869592 |
| Physical and Chemical Properties | amorphous powder. [α]D18 165 (C = 0.4, water). |
| Use | For the treatment of insulin-dependent and non-insulin-dependent diabetes mellitus |
56180-94-0 - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 1 |
| RTECS | LZ7153000 |
| HS Code | 29400090 |
| Toxicity | LD50 orl-rat: 24 g/kg NIIRDN -,2,1995 |
56180-94-0 - Nature
Open Data Verified Data
amorphous powder.
56180-94-0 - Use
Open Data Verified Data
oral hypoglycemic agents. Competitive inhibition of glucosidase in the intestinal tract can reduce the decomposition of polysaccharide and sucrose to produce glucose, reduce and delay absorption, thus reducing postprandial hyperglycemia and plasma insulin concentration, the product can competitively inhibit glucosidase in intestinal tract, reduce the decomposition of polysaccharide and sucrose to produce glucose, reduce and delay its absorption, and has the effect of reducing postprandial hyperglycemia and plasma insulin concentration.
56180-94-0 - Standard
Authoritative Data Verified Data
This product is 0-4, 6-dideoxy-4-[(1S,4R,5S,6S)-4,5, 6-trihydroxy-3-(hydroxymethyl) cyclohexen-2-yl] amino; a-D-glucopyranosyl-(1-4)-0-a-D glucopyranosyl-(1-4)-D-glucopyranose. The content of C25H43N018 shall be between 95.0% and 102.0%, calculated as anhydrous.
56180-94-0 - Trait
Authoritative Data Verified Data
- This product is white to light yellow amorphous powder, odorless.
- This product is very soluble in water, dissolved in methanol, very slightly soluble in ethanol, insoluble in acetone or acetonitrile.
specific rotation
take this product, precision weighing, water dissolution and quantitative dilution of about 5mg per lml solution, according to the law (General 0621), specific rotation of 168 ° to 183 °.
56180-94-0 - Differential diagnosis
Authoritative Data Verified Data
- in the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the reference solution.
- The infrared absorption spectrum of this product should be consistent with that of the reference product (General rule 0402).
56180-94-0 - Exam
Authoritative Data Verified Data
pH
take this product, add water to dissolve and make a solution containing 20mg per lml, according to the law (General 0631),pH value should be 5.5~7.5.
absorbance
take this product, add water to dissolve and dilute to make a solution containing about 50mg per 1 ml, and measure absorbance at the wavelength of 425mn by UV-visible spectrophotometry (General rule 0401), not more than 0.15.
Related substances
take an appropriate amount of this product, accurately weigh it, add water to dissolve and dilute it to make a solution containing about 20mg per lml as a test solution; Take 1ml for precision measurement and put it in a 100ml measuring flask, dilute to the scale with water, shake, as a control solution; Take the appropriate amount of control solution, diluted with water to make a solution containing about 10ug per 1ml, as a sensitivity solution. According to the chromatographic conditions under the content determination item, the sensitive solution 10ul is injected into the human liquid chromatograph, and the chromatogram is recorded. The signal-to-noise ratio of acarbose peak should be greater than 10; LOLs of the test solution and the control solution were respectively injected into the liquid chromatograph, and the chromatogram was recorded to 2.5 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution, the impurity content shall be calculated by the principal component self-control method multiplied by the correction factor, and shall conform to the corresponding limits in the table. Impurity peaks with a content of less than 0.05% are negligible.
moisture
take this product, according to the moisture determination method (General 0832 first method 1), the water content shall not exceed 4.0%.
ignition residue
take l.Og of this product and determine it according to law (General rule 0841). The remaining residue shall not exceed 0.2%.
Heavy metals
The residue left under the item of taking the ignition residue shall not contain more than 20 parts per million of heavy metal when examined by law (General rule 0821, Law II).
56180-94-0 - Content determination
Authoritative Data Verified Data
measured by high performance liquid chromatography (General 0512).
chromatographic conditions and system suitability test
with amino-bonded silica gel as filler (Welch Ultimate XB-NH2 column, mm x mm,5um or equivalent column); With phosphate buffer (take potassium dihydrogen phosphate 600mg and anhydrous disodium hydrogen phosphate mg, water was added to dissolve and diluted to 1000ml)-acetonitrile (25:75) as mobile phase; Flow rate was 2.0 mL per minute; Detection wavelength was 210nm; Column temperature was 35°C. Take about 200mg acarbose, put it in a 10ml measuring flask, add a small amount of water to dissolve, add 1ml of 0.1mol/L sodium hydroxide solution, mix well, place it at room temperature for 1 hour, add 0. 1ml of 1mol/L hydrochloric acid solution, diluted to scale with water, shake well, as the system applicable solution, take 10u1 injection liquid chromatograph, record chromatogram, the retention time of impurity I peak relative to acarbose peak is about 0.9, and the peak height of impurity I (HP, from baseline to the highest point of impurity I Peak) the ratio (Hp/Hv) to the peak-to-valley (Hv, from the baseline to the nadir between the two peaks) between the peaks of impurity I and acarbose shall not be less than 2.0, the number of theoretical plates shall not be less than 2000 based on the acarbose peak.
assay
take an appropriate amount of this product, accurately weigh it, add water to dissolve it and quantitatively dilute it to make a solution containing about 1 mg per lml, as a test solution, take 10u1 injection of human liquid chromatography with precise amount, the chromatogram was recorded, and an appropriate amount of acarbose reference substance was taken and determined by the same method. According to the external standard method to calculate the peak area, that is.
56180-94-0 - Category
Authoritative Data Verified Data
hypoglycemic agents.
56180-94-0 - Storage
Authoritative Data Verified Data
sealed and kept in cool and dark place.
56180-94-0 - Acarbose Tablets
Authoritative Data Verified Data
This product contains acarbose (C25H43N018) should be 95.0% to 105.0% of the label.
trait
This product is a white or light yellow film.
identification
In the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the control solution.
examination
- Related substances take an appropriate amount of fine powder of this product (about 500mg of acarbose), put it in a 25ml measuring flask, add an appropriate amount of water, shake to dissolve the acarbose, dilute it to the scale with water, shake it well, filter it, take the continued filtrate as the test solution; Take 1ml of the precise amount, put it in a 100ml measuring flask, dilute it to the scale with water, shake it, and use it as the control solution; Take the appropriate amount of the control solution, A solution containing about 10ug of acarbose per 1ml was prepared by dilution with water as a sensitivity solution. According to the chromatographic conditions under the content determination item, the sensitivity solution 10u1 is injected into the human liquid chromatograph, and the chromatogram is recorded. The signal-to-noise ratio of acarbose peak height should be greater than 10; 10 u1 of the test solution and the control solution were respectively injected into the human liquid chromatograph, and the chromatogram was recorded to 2.5 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution (the chromatographic peaks before the relative retention time 0.2 are subtracted), the impurity content shall be calculated by the principal component self-control method multiplied by the correction factor, and shall conform to the corresponding limits in the table. Impurity peaks with a content of less than 0.05% are negligible.
- the dissolution of this product, according to the dissolution and release determination method (General rule 0931 second method), water 900ml as the dissolution medium, the speed of 75 rpm, according to the law, after 30 minutes, take 10ml of the solution and filter, according to the chromatographic conditions under the content determination item (adjust the proportion of mobile phase so that the peak time of the main peak is between 5 and 10 minutes), the filtrate 30 u1 was taken from the precision child, and the human was injected into the liquid chromatograph, and the chromatogram was recorded, water was added to dissolve and quantitatively diluted to prepare a solution containing about 50ug(50mg specification) or 100 tons (lOOmg specification) per 1 ml, which was used as a reference solution and measured by the same method. According to the external standard method, the dissolution amount of each tablet is calculated from the peak area, and the limit is 80% of the labeled amount, which shall be in accordance with the provisions.
- others shall be in accordance with the relevant provisions under the item of tablets (General rule 0101).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using amino-bonded silica gel as filler (Welch ULtimate XB ^ NH2 column, 4.6mm X 250mm, 5um or equivalent column); phosphate buffer solution (600mg of potassium dihydrogen phosphate and 279mg of anhydrous disodium hydrogen phosphate, dissolved in water and diluted to 1000ml)-ethyl acetate (25:75) was used as mobile phase; The flow rate was 2.0 mL per minute; The detection wavelength was 210mn; column temperature 35°C. Take about 200mg acarbose, put it in a 10ml measuring flask, add a small amount of water to dissolve, add 0.1 mol/L sodium hydroxide solution 1ml, mix well, room temperature for 1 hour, add 0. 1ml of 1 mol/L hydrochloric acid solution, diluted to scale with water, shake well, as the system applicable solution, take 10u1 injection liquid chromatograph, record chromatogram, the retention time of impurity I peak to acarbose peak is about 0.9, and the peak of impurity I is high (HP, from baseline to the highest point of impurity I Peak) the ratio (Hp/Hv) to the peak-to-valley (Hv, from the baseline to the nadir between the two peaks) between the peaks of impurity I and acarbose shall not be less than 2.0. The number of theoretical plates shall not be less than 2000 based on the acarbose peak.
- determination Method: Take 20 tablets of this product, accurately weigh and grind, accurately weigh an appropriate amount of fine powder (about 50mg equivalent to acarbose), put it in a 50ml measuring flask, add an appropriate amount of water, and ultrasonic to dissolve acarbose, dilute with water to the scale, shake, filter, take the filtrate as the test solution, and inject 10ul into the liquid chromatograph to record the chromatogram, water was added and dissolved and diluted to prepare a solution containing about 1 mg per 1 ml, which was determined by the same method. According to the external standard method to calculate the peak area, that is.
category
Same as acarbose.
specification
(l)50mg (2)100mg
storage
sealed and kept in cool and dark place.
56180-94-0 - Acarbose capsules
Authoritative Data Verified Data
This product contains acarbose (C25H43N018) should be 95.0% to 105.0% of the label.
trait
The content of this product is white or white powder.
identification
In the chromatogram recorded under the content determination item, the retention time of the main peak of the test solution should be consistent with the retention time of the main peak of the control solution.
examination
- Related substances take an appropriate amount of the contents of this product (about 500mg of acarbose), put it in a 25ml measuring flask, add an appropriate amount of water, shake to dissolve the acarbose, dilute it to the scale with water, shake it well, filter it, take the continued filtrate as the test solution; Take 1 ml in a 100ml measuring flask, dilute with water to the scale, shake, and use as the control solution; Take the appropriate amount of the control solution, A solution containing about 10ug of acarbose per 1 ml was prepared by dilution with water as a sensitivity solution. According to the chromatographic conditions under the content determination item, the sensitive solution 10u1 is injected into the human liquid chromatograph, and the chromatogram is recorded. The signal-to-noise ratio of acarbose peak height should be greater than 10; l0ul of the test solution and the control solution were respectively injected into the human liquid chromatograph, and the chromatogram was recorded to 2.5 times of the retention time of the main component peak. If there are impurity peaks in the chromatogram of the test solution (the chromatographic peaks before the relative retention time 0.2 are subtracted), the impurity content shall be calculated by the principal component self-control method multiplied by the correction factor, and shall conform to the corresponding limits in the table. Impurity peaks with a content of less than 0.05% are negligible.
- the dissolution of this product, according to the dissolution and release determination method (General rule 0931 first method), water 900ml as the dissolution medium, the speed of 50 rpm, according to the law, after 30 minutes, take 10ml of the solution and filter it, and measure it according to the chromatographic conditions under the content determination item (adjust the mobile phase ratio so that the peak time of the main peak is between 5 and 10 minutes), the continuous filtrate 30 u1 was accurately weighed, and the liquid chromatograph was injected to record the chromatogram. The appropriate amount of acarbose reference substance was accurately weighed, dissolved by adding water and quantitatively diluted to make a solution containing about 50ug per 1 ml, as a control solution, the same method. According to the external standard method, the dissolution of each grain is calculated by the peak area, and the limit is 80% of the labeled amount, which should be in accordance with the provisions.
- moisture the contents of this product shall not contain more than 0832 of moisture as determined by the method for moisture determination (General rule 12.0%, first method 1).
- other requirements shall be in accordance with the relevant provisions under the paragraph of glue (General rule 0103).
Content determination
- measured by high performance liquid chromatography (General 0512).
- chromatographic conditions and system suitability test using amino-bonded silica as filler (Welch Ultimate XB-NH2 column, 4.6mm X 250mm, 5um or column h with equivalent performance using phosphate buffer solution (600mg of potassium dihydrogen phosphate and 279mg of anhydrous disodium hydrogen phosphate, dissolved in water and diluted to 1000ml)-acetonitrile (25:75) as mobile phase; the flow rate was 2.0 mL per minute; The detection wavelength was 210nm; And the column temperature was 35°C. Take about 200mg acarbose, put it in a 10ml measuring flask, add a small amount of water to dissolve, add 0.1 mol/L sodium hydroxide solution 1ml, mix well, place at room temperature for 1 hour, add O.lmol/L hydrochloric acid solution (lml), diluted to scale with water, shake well, as the system applicable solution, take 10u1 injection liquid chromatograph, record chromatogram, the retention time of impurity I relative to the acarbose peak is about 0.9, and the peak height of impurity I (Hp, from baseline to the highest point of impurity I Peak) the ratio (Hp/Hv) to the peak-to-valley (Hv, from the baseline to the nadir between the two peaks) between the peaks of impurity I and acarbose shall not be less than 2.0. The number of theoretical plates shall not be less than 2000 based on the acarbose peak.
- determination method: the content is appropriate under the difference of loading amount, mixed evenly, accurately weigh an appropriate amount (about 50mg equivalent to acarbose), place it in a measuring flask, add an appropriate amount of water, and sonicate the acarbose to dissolve, dilute with water to the scale, shake, filter, take the filtrate as the test solution, take 10u1 injection of human liquid chromatography with precise volume, record chromatogram; Take the appropriate amount of acarbose reference substance, precise weighing, water was added to dissolve and quantitatively diluted to prepare a solution containing about 1 mg per 1 ml, which was determined by the same method. According to the external standard method to calculate the peak area, that is.
category
Same as acarbose.
specification
50mg
storage
sealed and kept in cool and dark place.
Supplier List
CAS: 56180-94-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 56180-94-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 56180-94-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 56180-94-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 56180-94-0
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog
CAS: 56180-94-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 56180-94-0
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
CAS: 56180-94-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
CAS: 56180-94-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 56180-94-0
Tel: 0086-551-65418684
Email: sales@tnjchem.com
info@tnjchem.com
Mobile: 0086 189 4982 3763
QQ: 2881500840
Wechat: 0086 189 4982 3763
WhatsApp: 0086 189 4982 3763
Product List: View Catalog




