554-57-4
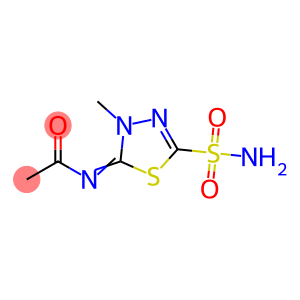
methazolamide
CAS: 554-57-4
Molecular Formula: C5H8N4O3S2
554-57-4 - Names and Identifiers
554-57-4 - Physico-chemical Properties
| Molecular Formula | C5H8N4O3S2 |
| Molar Mass | 236.27 |
| Density | 1.6625 (rough estimate) |
| Melting Point | 208 °C (dec.) (lit.) |
| Boling Point | 402.0±28.0 °C(Predicted) |
| Flash Point | 196.922°C |
| Water Solubility | 2.835g/L(25 ºC) |
| Solubility | DMSO: soluble20mg/mL, clear |
| Vapor Presure | 0mmHg at 25°C |
| Appearance | White powder |
| Color | white to beige |
| pKa | 7.30(at 25℃) |
| Storage Condition | 2-8°C |
| Refractive Index | 1.6270 (estimate) |
| Use | Used as a diuretic |
| In vitro study | Methazolamide is a carbonic anhydrase inhibitor with K I of 50 nM,14 nM, and 36 nM for hCA I,hCA II, and bCA IV subtypes, respectively. The advantage of Methazolamide is equivalent to acetazolamide, which is another carbonic anhydrase inhibitor for the treatment of irregular breathing disorders. However, methazolamide, unlike acetazolamide, does not activate Ca |
| In vivo study | In anaesthetized rabbits, Methazolamide did not impair respiratory function. |
554-57-4 - Risk and Safety
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R40 - Limited evidence of a carcinogenic effect |
| Safety Description | S22 - Do not breathe dust. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
| RTECS | AC6350000 |
| HS Code | 2935904000 |
554-57-4 - Reference Information
| NIST chemical information | information provided by: webbook.nist.gov (external link) |
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Introduction | methazolamide chemical name: 5-acetamido-4-methyl-1-thio-3, 4-diazocene sulfonamide, can be used to alleviate the high altitude reaction. |
| pharmacological effects | methazolamide is also a carbonic anhydrase inhibitor, mainly used in the treatment of glaucoma. Belongs to acetazolamide homolog, chemical structure similar to acetazolamide (one more methyl in the nitrogen atom), therefore, the pharmacological effect and mechanism of action and acetazolamide the same. The structural difference reduces the ionization effect, so the intraocular permeability is enhanced compared with acetazolamide. This product penetrates the blood-aqueous humor and blood-brain barrier is also stronger than acetazolamide (human cerebrospinal fluid concentration is 50 times higher than acetazolamide). Carbonic anhydrase inhibition is 60% stronger than acetazolamide, in vivo only 55% of methazolamide and plasma protein binding, so the lower dose is significantly lower intraocular pressure response. |
| indication | for primary open-angle glaucoma, angle-closure glaucoma and some secondary glaucoma, and adjuvant therapy for patients with poor intraocular pressure control by topical anti-glaucoma drugs. Because this product reduces intraocular pressure and has less effect on acid-base balance, it is better than acetazolamide for patients with severe obstructive pulmonary disease. The effect on urinary citrate secretion is smaller than that of acetazolamide, so for patients who need oral carbonic anhydrase inhibitor treatment but are easy to cause kidney stone formation, it is recommended to apply methazolamide. |
| adverse reactions | 1. It can cause serious hematological adverse reactions, including aplastic anemia and agranulocytosis. 2. May cause kidney stones, but very rare. 3. Other adverse reactions were Nausea, anorexia, paresthesia, discomfort, fatigue and skin erosion. |
| drug interaction | 1. Carbonic anhydrase inhibitors combined with high-dose aspirin can cause serious metabolic disorders, therefore, this product should be used with salicylic acid preparations with caution. 2. Low-dose methazolamide itself does not cause hypokalemia, but Carbonic anhydrase inhibitors can increase the potassium excretion effect of other drugs. 3. Combined use with adrenocorticotropic hormone and glucocorticoid can lead to severe hypokalemia, and attention should be paid to monitoring the concentration of serum potassium and cardiac function in combination. It should also be estimated that long-term concomitant use may increase the risk of hypocalcemia and may result in osteoporosis because these drugs increase calcium excretion. |
| biological activity | Methazolamide (CL 8490) is a carbonic anhydrase inhibitor targeting hCA I, for the hCA II and bCA IV subtypes, Ki values were 50 nM, 14 nM and 36 nM, respectively. |
| Target | Value |
| hCAII | 14 nM(Ki) |
| bCAIV | 36 nM(Ki) |
| hCAI | 50 nM(Ki) |
| Use | use as a diuretic |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Methazolamide Visit Supplier Webpage Request for quotationCAS: 554-57-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Methazolamide Request for quotation
CAS: 554-57-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 554-57-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Methazolamide Visit Supplier Webpage Request for quotation
CAS: 554-57-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 554-57-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Methazolamide Visit Supplier Webpage Request for quotationCAS: 554-57-4
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: Methazolamide Request for quotation
CAS: 554-57-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 554-57-4
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: Methazolamide Visit Supplier Webpage Request for quotation
CAS: 554-57-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 554-57-4
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


