5472-70-8
5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one
CAS: 5472-70-8
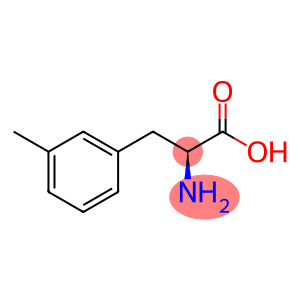
Molecular Formula: C10H13NO2
5472-70-8 - Names and Identifiers
| Name | 5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one |
| Synonyms | Nsc29447 3-Methylphe DL-3-Me-Phe-OH 3-Methy-DL-Phenylalanine DL-3-METHYLPHENYLALANINE 3-Methyl-DL-phenylalanine 2-AMINO-3-M-TOLYL-PROPIONIC ACID 3- Methylphenylalanine(3-Methylphe) 6H-Anthra[1,9-cd]isoxazol-6-one, 5-(cyclohexylamino)- 5-(Cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one 5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one |
| CAS | 5472-70-8 |
| InChI | InChI=1/C20H18N2O2/c23-19-13-8-4-5-9-14(13)20-18-16(22-24-20)11-10-15(17(18)19)21-12-6-2-1-3-7-12/h4-5,8-12,21H,1-3,6-7H2 |
5472-70-8 - Physico-chemical Properties
| Molecular Formula | C10H13NO2 |
| Molar Mass | 179.22 |
| Density | 1.165 |
| Melting Point | 245℃ |
| Boling Point | 322.8±30.0 °C(Predicted) |
| Flash Point | 296.8°C |
| Vapor Presure | 6.99E-13mmHg at 25°C |
| pKa | 2.22±0.10(Predicted) |
| Storage Condition | Keep in dark place,Inert atmosphere,Room temperature |
| Refractive Index | 1.712 |
5472-70-8 - Introduction
5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one(5-(cyclohexylamine)-6H-anthracene [1,9-cd][1,2]oxazol-6-one) is an organic compound whose chemical structure contains a methylphenylalanine group and a cyclohexylamine group.
The properties of the compound are as follows:
-Appearance: Colorless solid
-Melting point: about 180-182°C
-Solubility: Soluble in common organic solvents such as dimethyl sulfoxide and dichloromethane
5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one has certain application value in the field of pharmacy and biochemistry, including the following uses:
-As a drug intermediate: It can be used to synthesize and prepare some drugs, such as anti-cancer drugs and antibacterial drugs.
-As a chemical probe: It can be used to study interactions and reaction processes in biological systems.
Regarding the preparation method of 5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one, there are many methods to choose from, but the commonly used methods include:
-First, the phenylacetone is converted into phenylpropionate through a chemical reaction.
-Then, the phenylpropionate is reacted with an amine to generate the corresponding phenylalanine ester.
-Finally, through reactions such as hydrogenation and acid hydrolysis, the acylphenylalanine ester is converted to 5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one.
There is no clear research report on the safety information of the 5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one. However, as a general rule when handling chemicals, the following precautions should be observed when using this compound:
-Appropriate personal protective equipment, such as laboratory gloves and safety glasses, must be worn.
-Be careful to avoid contact with skin, eyes and mucous membranes during use and preparation.
-Before operation, you should be carefully familiar with the physical properties of the compound and the method of experimental operation.
-Avoid mixing or use with other incompatible chemicals.
Although the above information is based on existing literature reports, always follow laboratory safety procedures when using chemicals, and consult the latest and reliable safety data sheets and research reports before specific operations to ensure operational safety.
The properties of the compound are as follows:
-Appearance: Colorless solid
-Melting point: about 180-182°C
-Solubility: Soluble in common organic solvents such as dimethyl sulfoxide and dichloromethane
5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one has certain application value in the field of pharmacy and biochemistry, including the following uses:
-As a drug intermediate: It can be used to synthesize and prepare some drugs, such as anti-cancer drugs and antibacterial drugs.
-As a chemical probe: It can be used to study interactions and reaction processes in biological systems.
Regarding the preparation method of 5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one, there are many methods to choose from, but the commonly used methods include:
-First, the phenylacetone is converted into phenylpropionate through a chemical reaction.
-Then, the phenylpropionate is reacted with an amine to generate the corresponding phenylalanine ester.
-Finally, through reactions such as hydrogenation and acid hydrolysis, the acylphenylalanine ester is converted to 5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one.
There is no clear research report on the safety information of the 5-(cyclohexylamino)-6H-anthra[1,9-cd][1,2]oxazol-6-one. However, as a general rule when handling chemicals, the following precautions should be observed when using this compound:
-Appropriate personal protective equipment, such as laboratory gloves and safety glasses, must be worn.
-Be careful to avoid contact with skin, eyes and mucous membranes during use and preparation.
-Before operation, you should be carefully familiar with the physical properties of the compound and the method of experimental operation.
-Avoid mixing or use with other incompatible chemicals.
Although the above information is based on existing literature reports, always follow laboratory safety procedures when using chemicals, and consult the latest and reliable safety data sheets and research reports before specific operations to ensure operational safety.
Last Update:2024-04-09 21:54:55
Supplier List
Spot supply
Product Name: 2-Amino-3-(m-tolyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 5472-70-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-AMINO-3-M-TOLYL-PROPIONIC ACID Request for quotation
CAS: 5472-70-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5472-70-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-AMINO-3-M-TOLYL-PROPIONIC ACID Visit Supplier Webpage Request for quotation
CAS: 5472-70-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5472-70-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: 2-Amino-3-(m-tolyl)propanoic acid Visit Supplier Webpage Request for quotationCAS: 5472-70-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: 2-AMINO-3-M-TOLYL-PROPIONIC ACID Request for quotation
CAS: 5472-70-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 5472-70-8
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Product Name: 2-AMINO-3-M-TOLYL-PROPIONIC ACID Visit Supplier Webpage Request for quotation
CAS: 5472-70-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 5472-70-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


