546-43-0
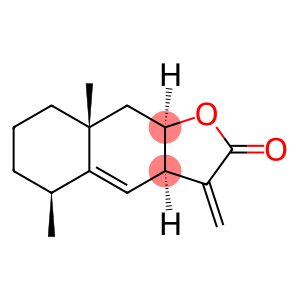
Alantolactone
CAS: 546-43-0
Molecular Formula: C15H20O2
546-43-0 - Names and Identifiers
546-43-0 - Physico-chemical Properties
| Molecular Formula | C15H20O2 |
| Molar Mass | 232.32 |
| Density | 1.0610 (rough estimate) |
| Melting Point | 78-79 C |
| Boling Point | 275 C |
| Specific Rotation(α) | D +175° (chloroform) |
| Flash Point | 111.5°C |
| Solubility | Soluble in ethanol, chloroform, benzene, ether and oil, almost insoluble in water. |
| Vapor Presure | 0.00523mmHg at 25°C |
| Appearance | Prismatic crystals (dilute ethanol) |
| Color | white to beige |
| Storage Condition | -20°C |
| Sensitive | Avoid light |
| Refractive Index | 1.4360 (estimate) |
| MDL | MFCD00274568 |
| Physical and Chemical Properties | Soluble in ethanol, chloroform, benzene, ether and oil, almost insoluble in water. From the root of Asteraceae Inula helenium L., the leaf of large complex flower Inula grandis chrenk. |
546-43-0 - Risk and Safety
| Hazard Symbols | Xi - Irritant |
| Risk Codes | R36/37/38 - Irritating to eyes, respiratory system and skin. R43 - May cause sensitization by skin contact |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S36/37 - Wear suitable protective clothing and gloves. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| HS Code | 29322090 |
546-43-0 - Reference
| Reference Show more | 1. Rezeng, Caidan, et al. "Alantolactone exhibited anti-herpes simplex virus 1 (HSV-1) action in vitro." Bioscience trends 9.6 (2015): 420-422.https://doi.org/10.5582/bst.2015.01171 2. [IF=5.64] Wu Dousheng et al."Oleanolic Acid Induces the Type III Secretion System of Ralstonia solanacearum."Front Microbiol. 2015 Dec;0:1466 3. [IF=4.556] Yushuang Ding et al."Induction of ROS Overload by Alantolactone Prompts Oxidative DNA Damage and Apoptosis in Colorectal Cancer Cells."Int J Mol Sci. 2016 Apr;17(4):558 4. [IF=2.4] Caidan Rezeng et al."Alantolactone exhibited anti-herpes simplex virus 1 (HSV-1) action in vitro."BioScience Trends. 2016 Jan 18 5. [IF=7.971] Hongge Wang et al."Synergistic lethality between PARP-trapping and alantolactone-induced oxidative DNA damage in homologous recombination-proficient cancer cells."Oncogene. 2020 Feb;39(14):2905-2920 |
546-43-0 - Reference Information
| Overview | Inula helenium L. is a perennial herb of the genus Inula of the Compositae family. It is mainly produced in Hebei. In addition, it is also produced in Zhejiang, Sichuan, Henan, Shanxi, Shaanxi, Gansu and Xinjiang. Its root is used for medicinal purposes. It has the effects of invigorating the spleen and stomach, regulating qi and relieving depression, relieving pain and preventing fetus. It is used for chest and hypochondrium, abdominal distension and pain, vomiting and diarrhea, chest and hypochondrium contusion, bifurcation and pain, and fetal movement. Modern pharmacology has proved that the active component of its volatile oil, alantolactone, has good deworming effect, antibacterial effect, and has exciting and inhibitory effects on cardiovascular diseases. Isobalactone (isoalantolactone) also has good deworming effect. There have been reports on the isolation of enoseolactone and isoseolactone in Auschyla. The author uses the different positions of the double bonds of soil and soil, and the firmness of the complex formed with Ag is different, which can separate the two components. Therefore, two single crystal components were obtained by Ag complex column chromatography from acetone soluble part of Solanum fructus. According to their physical and chemical constants and spectral data analysis, they were identified as alantolac-tone (I) and isoalantolactone (II) respectively. Studies have found that acetone is better than petroleum ether in the extraction effect of enoseolactone and isoseolactone. At the same time, this process is easy to carry out large-scale industrial production [3]. |
| determination of content | GC method (1) chromatographic conditions chromatographic column: carrier, Chromsorb W-HP (80~100 mesh); Fixing solution, 3 AP (reactant of polyethylene glycol 2mol/L and 2-nitrobenzoic acid);(2m × 2mm) stainless steel column; Column temperature: 158 ℃; Injector temperature; 200 ℃; detector: FID; Carrier gas: N2 flow rate 30 ml/min;H2 flow rate 0.6 ml/min; Air pressure: 49.03kPa. (2) preparation of reference substance and internal standard solution precisely weigh 11.11mg of reference substance, place it in a 1ml volumetric flask, add chloroform to dissolve, and prepare standard reference substance reserve solution. In addition, precisely weigh 5.7mg of 23 alkane, place it in a 1ml volumetric flask, add chloroform to the scale, and be the internal standard reserve solution. (3) for determination of correction factor, use a 100 μl microsyringe to suck 160 μl of standard reference substance stock solution and place it in a 1ml measuring flask, then add 100 μl of internal standard stock solution into the measuring flask, add chloroform to scale, mix evenly, inject 1μl into a gas chromatograph, record chromatogram, and calculate correction factor with peak area. (4) preparation of sample solution: take about 1g of wood incense powder, weigh it accurately, place it in a stopper bottle, accurately add 3ml of chloroform, cold soak overnight, take appropriate amount of filtrate, add 120 μl of internal standard solution, fix the volume of chloroform to 1ml as sample solution. (5) determine 1 μl of sample solution, inject it into gas chromatograph, and calculate the percentage content of soil and soil lactone in medicinal materials according to internal standard method. (6) chromatogram fig. 1 gas chromatogram of reference substance and internal standard 1. internal standard substance, tR = 6min; 2. enoseolactone, tR = 13min; 3. iso-enoseolactone, TR = 19min [effect on proliferation and apoptosis of RPMI-8226 cells of multiple myeloma] [2] RPMI-8226 cells were treated with aconutinolactone with concentrations of 1, 2. 5, 7. 5 and 10 μmol/L for 48 h, cell viability was detected by CCK-8, and IC50 value was calculated. The concentration used was 2. 5, after treating RPMI-8226 cells with 5 and 7. 5 μmol/L enoseolactone for 48 h, apoptosis was analyzed by Annexin V/PI double labeling method. Blot method was Western to detect the changes of cleaved caspase-3 and ERK signaling pathway. Nude mice were bearing tumor and given enoseolactone to further verify its apoptosis-promoting effect on multiple myeloma cells. The IC50 of RPMI-8226 cells at 48 h was found to be 4.32±0. 15 μmol / L; flow cytometry showed that the rate of early apoptosis increased significantly with the increase of the concentration of the cells (r = 0. 9784,P = 0. 0007); western blot results showed that after the treatment of RPMI-8226 cells, the caspase-3 was activated and ERK phosphorylation level was reduced. In vivo results showed that the tumor volume of mice in the administration group was significantly smaller than that in the control group (P <0. 05). Immunohistochemical test results showed that compared with the control group, the levels of nuclear proliferation-related antigen Ki67 and p-ERK in the experimental group decreased. Conclusion: Enoseolactone can effectively inhibit the proliferation and promote the apoptosis of multiple myeloma RPMI-8226 cells, and its mechanism may be to inhibit the growth of subcutaneous multiple myeloma in nude mice by inhibiting the activity of ERK signaling pathway. Previous studies have found that enagrolactone has a variety of anti-tumor effects. In this study, RPMI-8226 cells of multiple myeloma were selected as the research object, and the anti-tumor effect of Enoseolactone on multiple myeloma was studied in vitro and in vivo. Firstly, CCK-8 was used to detect the killing effect of Enoseolactone on RPMI-8226 cells, and it was found that it could significantly inhibit the cell activity of myeloma, with 48 hIC50 being 4. 32±0. 15 μmol/L. Previously, Mi et al. reported that human normal stem cell line L02 was less sensitive to enoseolactone, and its IC50 was about 31. 34 μg /ml( 133.75 μmol/L). It can be seen that the killing effect of Enoseolactone on normal cells is low, and the killing effect on myeloma cells is obvious at a lower concentration. Further flow cytometry was used to detect the treatment of myeloma cells with enagrolactone. It was found that enagrolactone played an anti-myeloma role by inducing apoptosis. |
| separate and extract | 50 kg of earthwood slices, and extract four times with acetone percolation. After recovering the solvent, the light yellow oil is obtained, placed at -5 ℃ and left to stand overnight, crystallized, and filtered to obtain 250 kg of light yellow needle crystal (mixture of Ⅰ and Ⅱ). 100 g of the crystal was taken, separated repeatedly by alumina-silver nitrate column chromatography and silica gel-silver nitrate column chromatography, eluted with petroleum ether-benzene-ethyl acetate mixed solvent, detected by thin layer chromatography, and the same parts of thin layer spots were collected and merged. The thin layer is detected as Ⅰ part of the recovered solvent and then dissolved with a small amount of ethyl acetate, then a large amount of petroleum ether is added to obtain crystal Ⅰ(16g), the thin layer is detected as Ⅱ part of the recovered solvent and then recrystallized with ethyl acetate to obtain Ⅱ(20g). As a traditional Chinese medicine with clear medicinal effects, it has been used to replace Aristolochic acid-containing Aristolochic acid in the prescription of patent medicine in recent years. The research on the active components of Aoxylenolactone and Aoxylenolactone in its volatile oil has also received increasing attention. Using acetone percolation and repeated separation by alumina-silver nitrate column chromatography and silica gel-silver nitrate column chromatography, pure products of enoseolactone and isoseolactone were obtained. This method has high product yield and simple operation, and can be used in the large-scale production process of soil and soil. Since silver nitrate is easy to decompose when it comes to light, measures should be taken to avoid light in this production process. |
| References | [1] Sun Wenji, edited by Xie Shichang; edited by Zhao Ye, Zhang Xingqun, Chen Qianliang, etc. Quantitative analysis of natural pharmaceutical ingredients. Beijing: China Medical Science and Technology Press. 2003. Page 12-13. [2] Yao Yao, Sun Yueyue, Xia Dandan, Niu Mingshan, Zhao Kai, Li Zhenyu, Zeng Lingyu, Xu Kailin. Inhibition of RPMI-8226 Cell Proliferation and Its Related Mechanisms [J]. Chinese Journal of Experimental Hematology, 2015,23(05):1336-1340. [3] Liu Guoyu, Li Jing, Wang Xinli, Zhou Junhui, wang Daqi. Isolation and Identification of Eupholactone and Eupholactone from Eupholactone [J]. Shaanxi Agricultural Sciences, 2011,57(04):9-10. |
| use | used for content determination/identification/pharmacological experiments, etc. Pharmacological effects: 1. Insect repellent: The effect is similar to that of Shan Dao Nian. It also has an insect repellent effect on pig roundworms and cat parasites. 2. Antibacterial: In vitro experiments prove that the growth of Mycobacterium tuberculosis can be inhibited at the concentration of 0.1 μg/ml. Administration can delay the onset of guinea pigs infected with human Mycobacterium tuberculosis, but it cannot be completely stopped. In addition, there is a strong antifungal effect. 3. Others: low concentration of drugs can excite the isolated frog heart, while higher concentration can inhibit and stop it in diastole. When the blood vessels of the frog hind limbs and rabbit ears are perfused, the low concentration of the drug has a slight dilation effect, and the high concentration shrinks. It also has a certain effect on the blood pressure and respiration of animals. |
Last Update:2024-04-09 19:05:12
Supplier List
Product Name: Alantolactone Request for quotation
CAS: 546-43-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 546-43-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Alantolactone Visit Supplier Webpage Request for quotationCAS: 546-43-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Multiple SpecificationsSpot supply
Product Name: (3aR,5S,8aR,9aR)-5,8a-Dimethyl-3-methylene-3,3a,6,7,8,8a,9,9a-octahydronaphtho[2,3-b]furan-2(5H)-one Visit Supplier Webpage Request for quotationCAS: 546-43-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Alantolactone Visit Supplier Webpage Request for quotationCAS: 546-43-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: Alantolactone Request for quotation
CAS: 546-43-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 546-43-0
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: Alantolactone Visit Supplier Webpage Request for quotationCAS: 546-43-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Multiple SpecificationsSpot supply
Product Name: (3aR,5S,8aR,9aR)-5,8a-Dimethyl-3-methylene-3,3a,6,7,8,8a,9,9a-octahydronaphtho[2,3-b]furan-2(5H)-one Visit Supplier Webpage Request for quotationCAS: 546-43-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Alantolactone Visit Supplier Webpage Request for quotationCAS: 546-43-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



